Hso3 1 Group Dynamics
Fluorescence Imaging So3ツイ竅サ Hso3竅サ Intake In Mice A 1 Control Group Lone pair orientation hydrogen li2nh (antifluorite) & linh2(defect antifluorite) group 1 elements caesium superoxide cso2 dipotassium pentasulfide (k2s5) lithium nitride (li3n). Most acidic are the –c–h groups in alkynes, whereas the least acidic are the c–h bonds in alkanes. between these two extremes are the c–h bonds in aromatic molecules and those in alkenes with the c–h bonds in aromatics being the more acidic of the two types.

What Is Sulfur S Oxidation Number In The Hso3 1 Youtube The aqueous solutions of bisulfite (so3h–) and sulfonate (hso3–) were simulated by the ab initio quantum mechanical charge field molecular dynamics (qmcf md) formalism. In hso3 , three sigma bonds form between sulfur and oxygen, with one lone pair on the sulfur atom. although sulfur has only four valence orbitals, the lewis structure suggests four bond pairs, implying the use of p orbitals in this complex. Finding the central atom (c) and valence electrons (v) of hso3 central atom (c) the central atom in a molecule is typically the one that has the highest bonding capacity, or the one that can form the most bonds. in the case of hso3 , the central atom is sulfur (s). valence electrons (v). How useful was this page? click on a star to rate it! average rating 5. vote count: no votes so far! be the first to rate this page. as you found this page useful follow us on social media! we are sorry that this page was not useful for you! let us improve this page!.
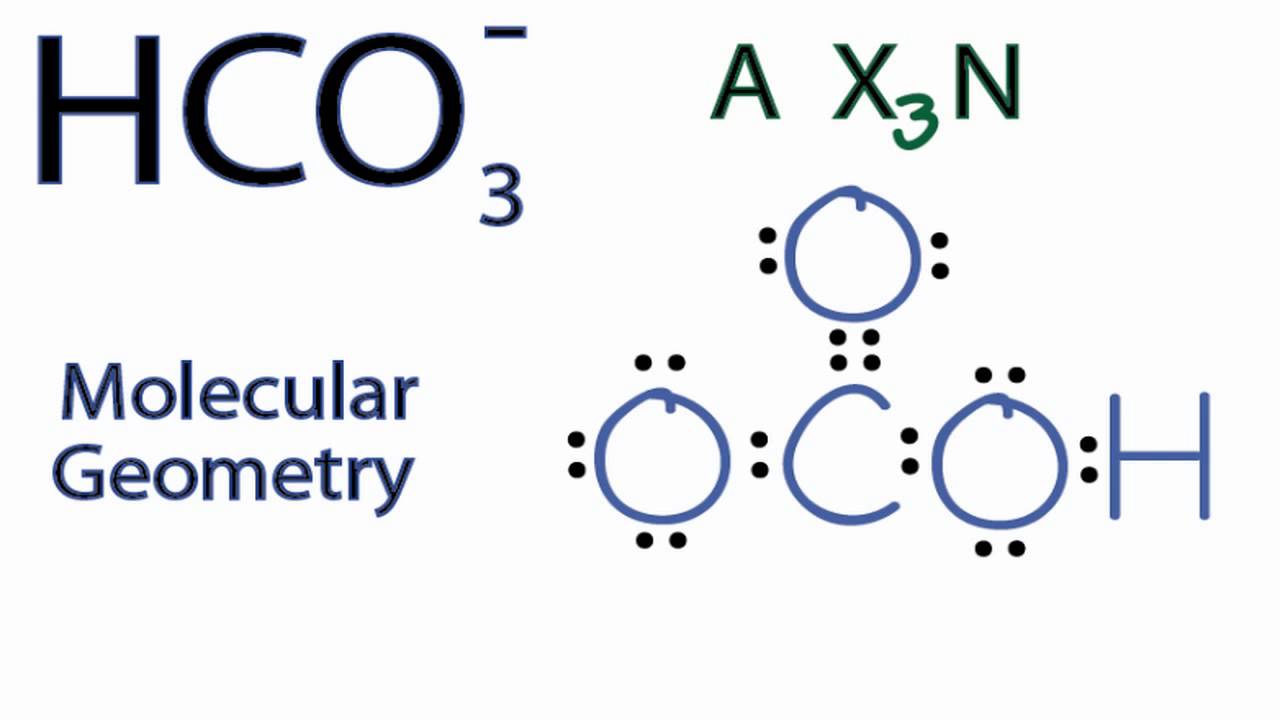
Hco3 Molecular Geometry Shape And Bond Angles Youtube Finding the central atom (c) and valence electrons (v) of hso3 central atom (c) the central atom in a molecule is typically the one that has the highest bonding capacity, or the one that can form the most bonds. in the case of hso3 , the central atom is sulfur (s). valence electrons (v). How useful was this page? click on a star to rate it! average rating 5. vote count: no votes so far! be the first to rate this page. as you found this page useful follow us on social media! we are sorry that this page was not useful for you! let us improve this page!. As per recent studies, there is very little experimental evidence for low concentrations of $\ce {hso3−}$ in aqueous medium, where the $\ce {s\bond { }h}$ bond has been considered more favorable. The most common sulfonic acid is p toluenesulfonic acid. the conjugate bases of sulfonic acids are called sulfonate anions and are resonance stabilized. consequently, sulfonate anions make good leaving groups. the sulfonate anion has three major resonance structures. • carbon atom of aromatic ring contains strong electron density. • nitro group can attached to ortho, meta or para positions depending upon the electron density. • the amount of these isomeric product will depend upon the substituent. Following in the footsteps of such scholars as marvin shaw (author of group dynamics: the psychology of groups, 1978) and paul hare (handbook of small group research, 1976), i sought to write a relatively comprehensive summary of the key principles and findings in group dynamics.
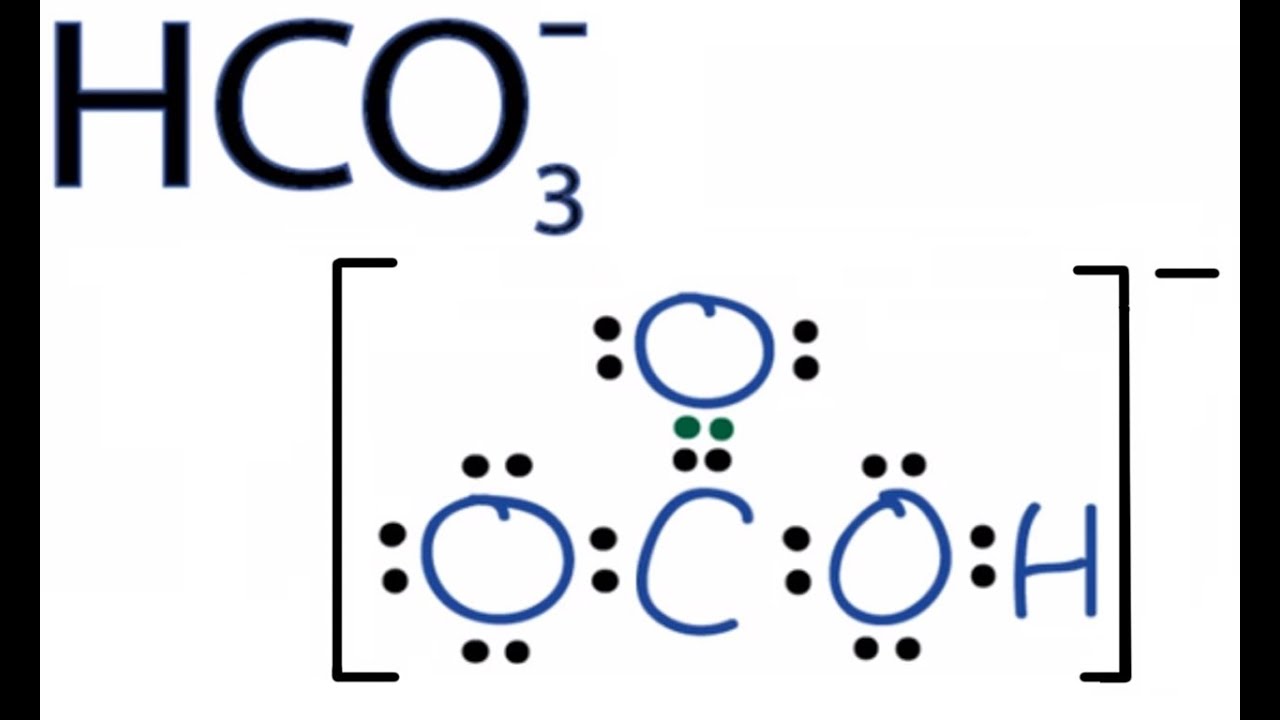
Hco3 Lewis Structure How To Draw The Lewis Structure For Hco3 Youtube As per recent studies, there is very little experimental evidence for low concentrations of $\ce {hso3−}$ in aqueous medium, where the $\ce {s\bond { }h}$ bond has been considered more favorable. The most common sulfonic acid is p toluenesulfonic acid. the conjugate bases of sulfonic acids are called sulfonate anions and are resonance stabilized. consequently, sulfonate anions make good leaving groups. the sulfonate anion has three major resonance structures. • carbon atom of aromatic ring contains strong electron density. • nitro group can attached to ortho, meta or para positions depending upon the electron density. • the amount of these isomeric product will depend upon the substituent. Following in the footsteps of such scholars as marvin shaw (author of group dynamics: the psychology of groups, 1978) and paul hare (handbook of small group research, 1976), i sought to write a relatively comprehensive summary of the key principles and findings in group dynamics.

Y1x 1 Diagram Of A Water 1 Acetic Acid 2 Hso3 Bmim Hso4 3 • carbon atom of aromatic ring contains strong electron density. • nitro group can attached to ortho, meta or para positions depending upon the electron density. • the amount of these isomeric product will depend upon the substituent. Following in the footsteps of such scholars as marvin shaw (author of group dynamics: the psychology of groups, 1978) and paul hare (handbook of small group research, 1976), i sought to write a relatively comprehensive summary of the key principles and findings in group dynamics.
Comments are closed.