How To Sample Unknown Liquids

Identifying Unknown Liquids A Density Lab Experiment Guide Course Hero This video is to help first responders and industrial hazmat teams with limited resources to identify an unknown liquid which could be a hazardous material. Procedure: into 4 labeled test tubes, add 1 ml of : water; 5% aq hcl; 5% aq nahco 3; and 5% aq. naoh. to each tube, add approx. 30 mg of the unknown. shake or stir for a few seconds. record your observations. it may take several minutes for the unknown to appear to dissolve or react.

Unknown Liquids Lab Unknown Liquids Problem 09 25 2015 Chem 2003 Lab Pipet 10.00 ml of the unknown liquid into the erlenmeyer flask. to review the techniques for using the pipette, the below link will open up a tutorial for using a pipette in a new window. Students test four known liquids and an unknown liquid on two different paper surfaces to investigate the question: can you identify liquids based on how they interact with different surfaces?. For the colorless liquid, tests include measuring density, melting point, boiling point, and solubility in water and ethanol. the purpose is to experimentally determine the identity of each unknown through comparing results to known substances. safety precautions are outlined for both experiments. Assume that 1 2 drops of liquid unknown is equal to 0.2 mmol or 20 mg. many of the reagents will have, attached to the bottle, pasteur pipets which are marked off at a certain volume. in other cases, measuring amounts by counting drops with a pipet calibrated in drops ml will be sufficient.

Ous Liquids Your Unknown Will Be One Of The Liquids Chegg For the colorless liquid, tests include measuring density, melting point, boiling point, and solubility in water and ethanol. the purpose is to experimentally determine the identity of each unknown through comparing results to known substances. safety precautions are outlined for both experiments. Assume that 1 2 drops of liquid unknown is equal to 0.2 mmol or 20 mg. many of the reagents will have, attached to the bottle, pasteur pipets which are marked off at a certain volume. in other cases, measuring amounts by counting drops with a pipet calibrated in drops ml will be sufficient. Unknown solids and visible material remaining from evaporated liquids may be analyzed by sem eds for elemental components. cient sample size may be suitable for xrd analysis. if necessary,. We provide you with a list of possible unknown compounds. (this situation reflects some real world organic laboratory synthetic situations, since often a chemist is working in a scheme and has a pretty good idea of the identity of an isolated compound.). Physical properties: identification of a pure liquid prepared by edward l. brown, lee university to identify an unknown liquid by comparing its measured physical properties (such as boiling point and density) with known values. Before making a refractive index measurement of the unknown liquid, your instructor should brief you on the proper operating procedure for the refractometer. next, for practice, determine the refractive index of distilled water.
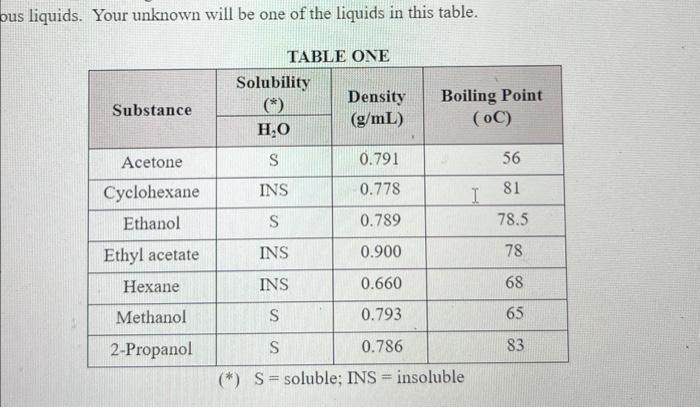
Ous Liquids Your Unknown Will Be One Of The Liquids Chegg Unknown solids and visible material remaining from evaporated liquids may be analyzed by sem eds for elemental components. cient sample size may be suitable for xrd analysis. if necessary,. We provide you with a list of possible unknown compounds. (this situation reflects some real world organic laboratory synthetic situations, since often a chemist is working in a scheme and has a pretty good idea of the identity of an isolated compound.). Physical properties: identification of a pure liquid prepared by edward l. brown, lee university to identify an unknown liquid by comparing its measured physical properties (such as boiling point and density) with known values. Before making a refractive index measurement of the unknown liquid, your instructor should brief you on the proper operating procedure for the refractometer. next, for practice, determine the refractive index of distilled water.
Comments are closed.