How To Name Acids Examples And Practice

Acids Definition Types Examples Properties Uses In these worksheets, students will dive deep into the rules and conventions for naming various acids, from the familiar ones found in everyday substances to more complex compounds encountered in advanced chemistry. Fill in the table by selecting the correct name for each formula. apply your understanding of naming acids and bases in this set of free, standards aligned practice questions.
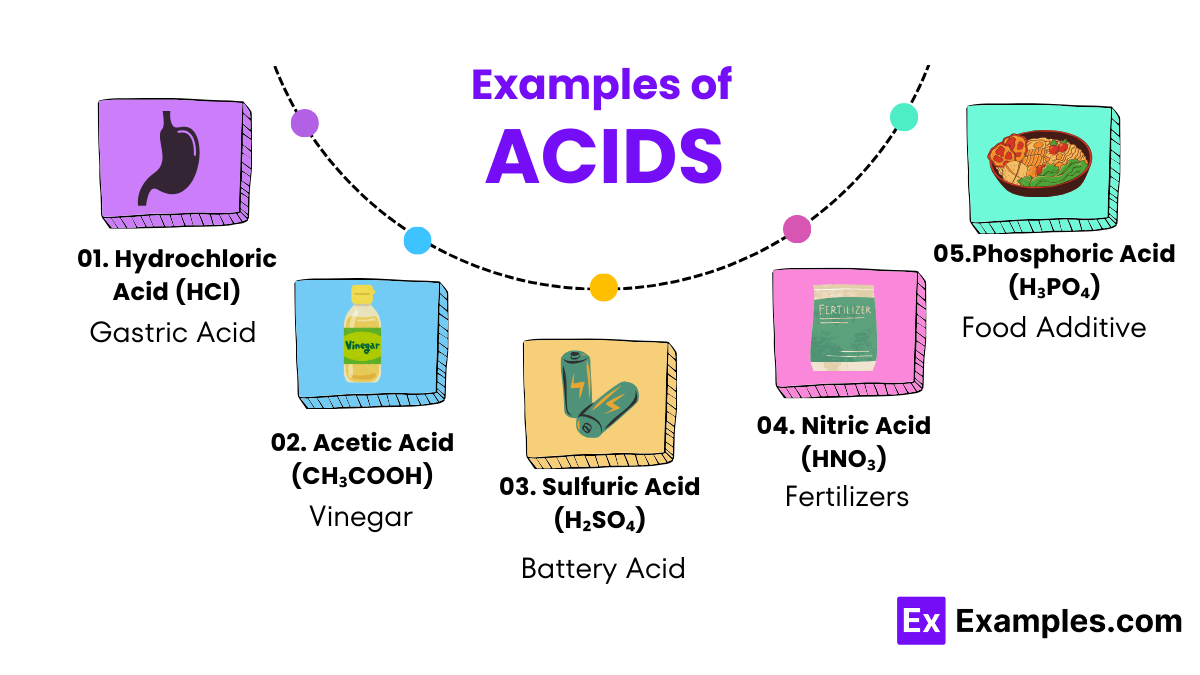
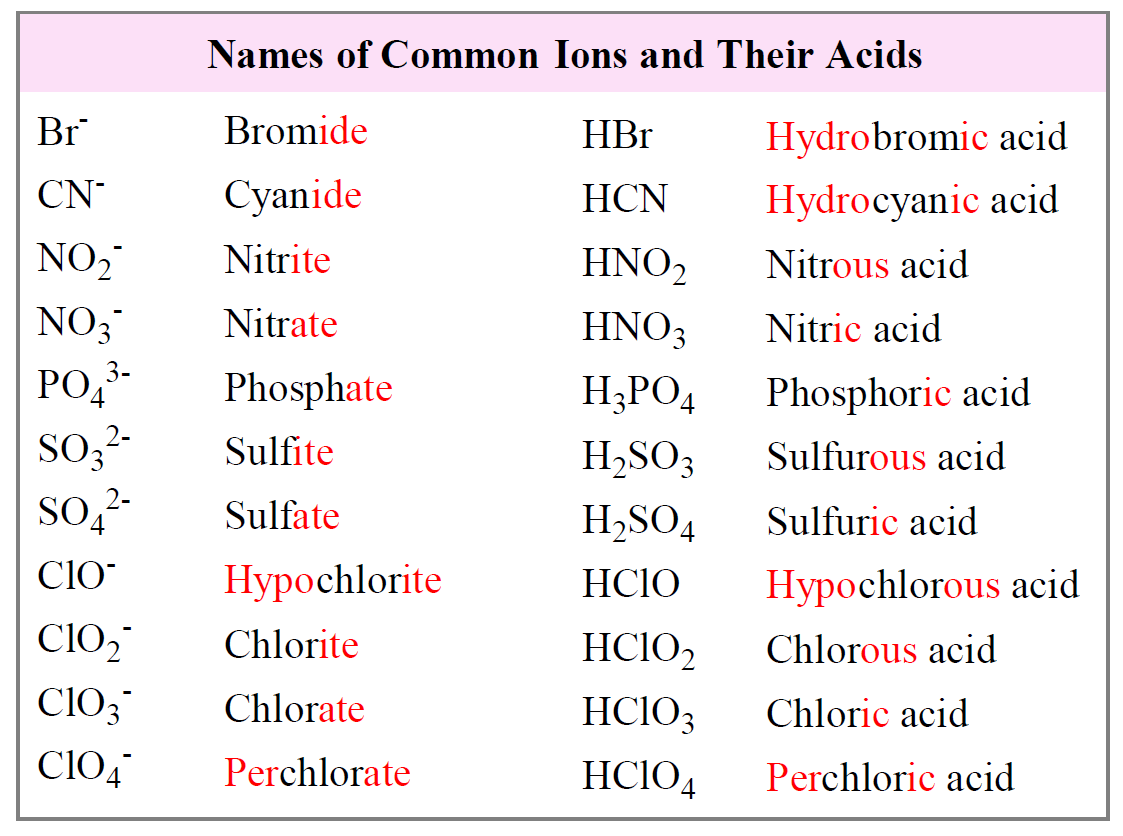
Acids Definition Types Examples Properties Uses This page titled acid nomenclature (worksheets) is shared under a cc by nc sa 4.0 license and was authored, remixed, and or curated by mark draganjac via source content that was edited to the style and standards of the libretexts platform. This practice quiz serves multiple instructional purposes and works effectively as a warm up activity to activate prior knowledge before introducing new acid base concepts, or as targeted practice after initial instruction on acid nomenclature. Although it is not always accurate, you can recognize acids when you see an h with any of the anions you learned so far. for example, hcl, h 2 so 4, hno 3, and h 3 po 4. The name for an acid is based on the name of the anion. if the anion ends with the letters –ide, the acid is named one way while acids containing anions that end with –ate use a different rule. remember that monatomic anions typically end with –ide. the rules for naming acids are summarized below.

How To Name Acids Examples And Practice Channels For Pearson Although it is not always accurate, you can recognize acids when you see an h with any of the anions you learned so far. for example, hcl, h 2 so 4, hno 3, and h 3 po 4. The name for an acid is based on the name of the anion. if the anion ends with the letters –ide, the acid is named one way while acids containing anions that end with –ate use a different rule. remember that monatomic anions typically end with –ide. the rules for naming acids are summarized below. Naming oxyacids: these are more difficult to name because these acids have hydrogen, a nonmetal, and may have varying numbers of oxygen atoms. for example, h2so5, h2so4, h2so3, and h2so2 are all acids. This document contains a worksheet with 22 questions focused on naming and identifying various acids and their formulas. it includes multiple choice questions about specific acids, their chemical formulas, and naming conventions for acids. An acid can be defined in several ways. the most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h ) when dissolved in water. At this very beginning level, you will recognize an acid by the fact that its formula starts with h, as in these examples:.
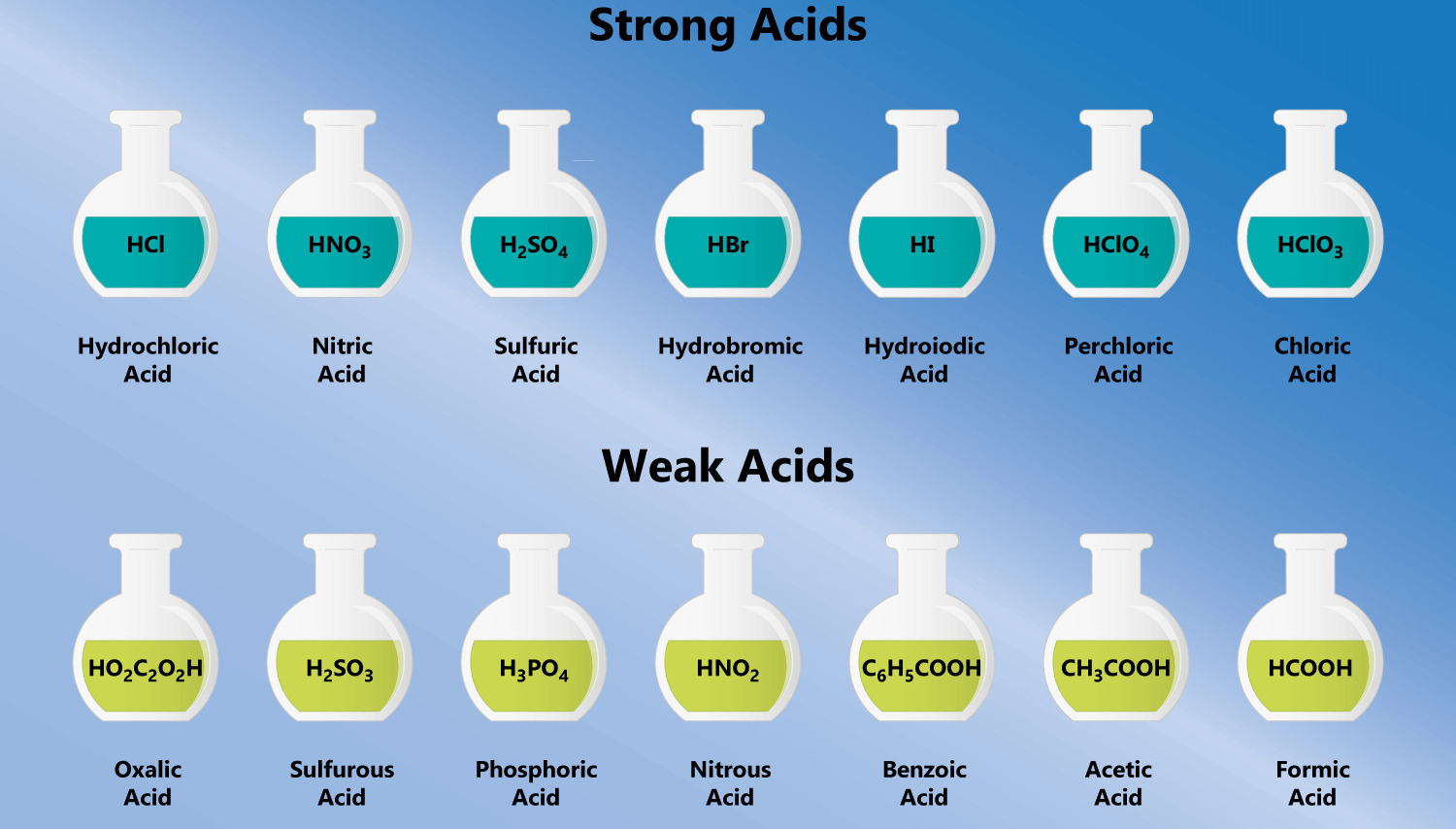
18 Facts About Acids You Should Know Facts Net Naming oxyacids: these are more difficult to name because these acids have hydrogen, a nonmetal, and may have varying numbers of oxygen atoms. for example, h2so5, h2so4, h2so3, and h2so2 are all acids. This document contains a worksheet with 22 questions focused on naming and identifying various acids and their formulas. it includes multiple choice questions about specific acids, their chemical formulas, and naming conventions for acids. An acid can be defined in several ways. the most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h ) when dissolved in water. At this very beginning level, you will recognize an acid by the fact that its formula starts with h, as in these examples:.
Naming Acids And Bases Chemistry Steps An acid can be defined in several ways. the most straightforward definition is that an acid is a molecular compound that contains one or more hydrogen atoms and produces hydrogen ions (h ) when dissolved in water. At this very beginning level, you will recognize an acid by the fact that its formula starts with h, as in these examples:.

Acids Naming Quiz Quiz Now
Comments are closed.