How Do Catalysts Work

How Catalysts Function Understanding Their Role In Reaction Rates And Catalysts function by allowing the reaction to take place through an alternative mechanism that requires a smaller activation energy. this change is brought about by a specific interaction between the catalyst and the reaction components. A catalyst changes how fast a chemical reaction happens by lowering the activation energy needed. positive catalysts make reactions go faster, while inhibitors slow them down or stop them. catalysts can work with different substances, like enzymes in the body or metals in cars.
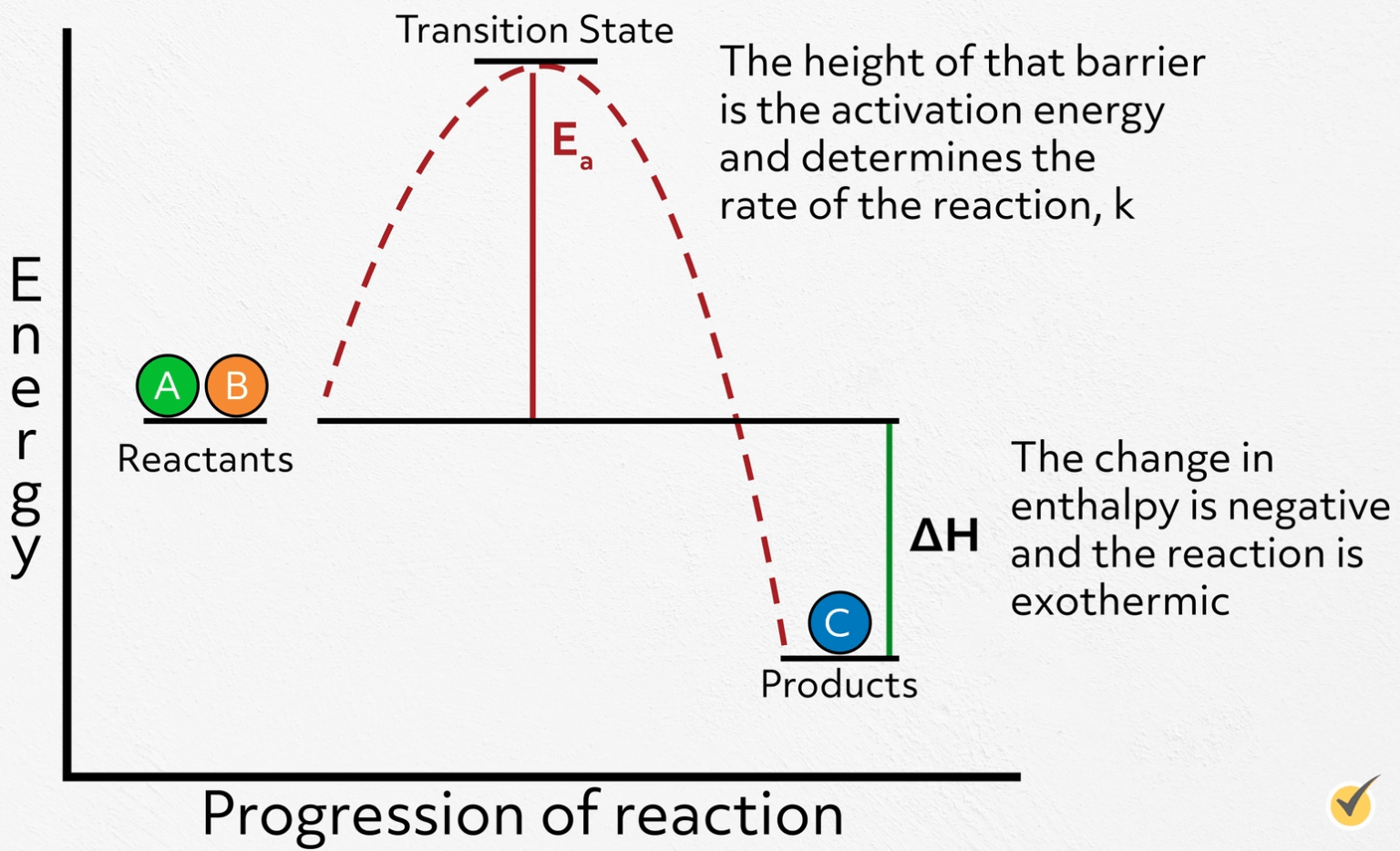
What Is A Catalyst Chemistry Review Video Learn how catalysts speed up or lower the energy barrier of chemical reactions, and how they can be designed to produce new materials and fuels. find out how doe supports basic research on catalysts and their applications in solar fuels, plastic upcycling, and more. It works by lowering the activation energy, the minimum energy needed for a reaction to get started, giving the reacting molecules an easier path from start to finish. the catalyst participates in intermediate steps along the way, but it emerges chemically unchanged once the reaction is complete. Learn the definition, types, and mechanisms of catalysts, substances that speed up chemical reactions without changing themselves. discover how catalysts shape the world of biology, industry, and the earth's atmosphere. A catalyst is a substance that increases the rate of a chemical reaction without being consumed by it. learn how catalysts work, examples of catalysts, types of catalysis, and units of catalysis.

Catalysts Activation Energy Chemtalk Learn the definition, types, and mechanisms of catalysts, substances that speed up chemical reactions without changing themselves. discover how catalysts shape the world of biology, industry, and the earth's atmosphere. A catalyst is a substance that increases the rate of a chemical reaction without being consumed by it. learn how catalysts work, examples of catalysts, types of catalysis, and units of catalysis. How do catalysts actually work? catalysts accelerate chemical reactions by providing alternative pathways with lower activation energy, forming transient intermediates, and modulating reactant properties, all without being consumed in the process. Catalytic reaction or catalysis is the process that alters the rate of a chemical reaction under the influence of a catalyst. a catalyst is a chemical substance added to a reaction to either accelerate or decelerate the reaction rate without itself undergoing any change. Catalyst, in chemistry, any substance that increases the rate of a reaction without itself being consumed. enzymes are naturally occurring catalysts responsible for many essential biochemical reactions. in general, catalytic action is a chemical reaction between the catalyst and a reactant. How catalysts work a catalyst provides an alternative reaction pathway that has a lower activation energy than the uncatalysed reaction. this does not change the frequency of collisions.
Solved Catalysts Work By The Activation Energy Of The Reaction How do catalysts actually work? catalysts accelerate chemical reactions by providing alternative pathways with lower activation energy, forming transient intermediates, and modulating reactant properties, all without being consumed in the process. Catalytic reaction or catalysis is the process that alters the rate of a chemical reaction under the influence of a catalyst. a catalyst is a chemical substance added to a reaction to either accelerate or decelerate the reaction rate without itself undergoing any change. Catalyst, in chemistry, any substance that increases the rate of a reaction without itself being consumed. enzymes are naturally occurring catalysts responsible for many essential biochemical reactions. in general, catalytic action is a chemical reaction between the catalyst and a reactant. How catalysts work a catalyst provides an alternative reaction pathway that has a lower activation energy than the uncatalysed reaction. this does not change the frequency of collisions.
Comments are closed.