Histone Acetylation Youtube
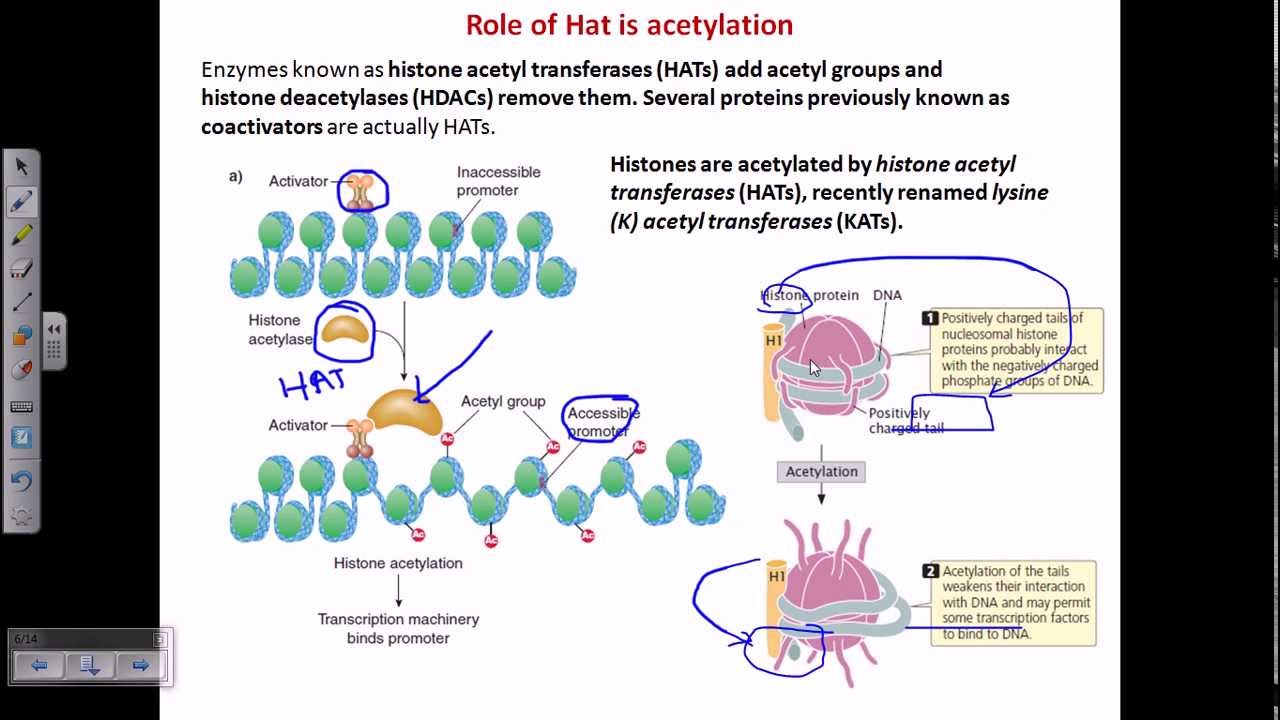
Histone Acetylation Youtube By the end of this short video, you'll understand exactly how histone acetylation works to control which parts of your dna are active. hope this animated explanation made things click for you!. Histone acetylation alters chromatin structure. shown in this illustration, the dynamic state of histone acetylation deacetylation regulated by hat and hdac enzymes.
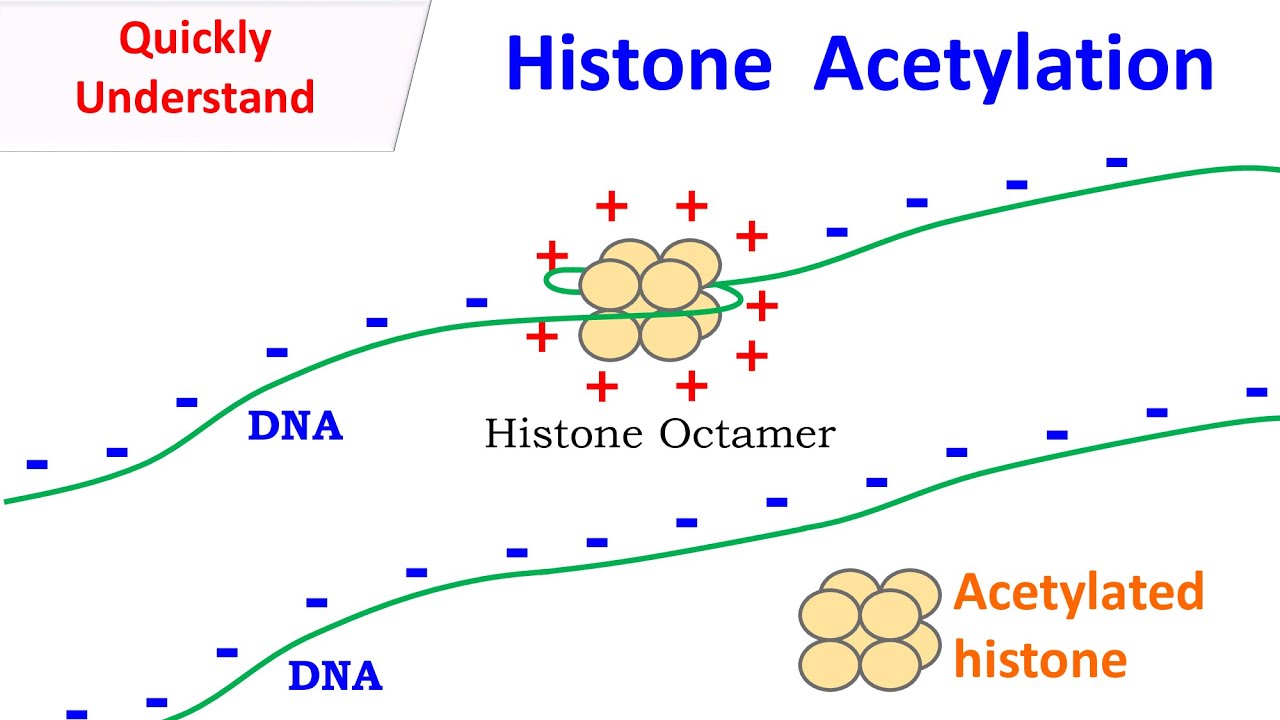
Histone Acetylation Youtube The histone proteins have a flexible n terminal tail extending out from the nucleosome. these histone tails are often subjected to post translational modifications such as acetylation, methylation, phosphorylation, and ubiquitination. This tutorial about chromatin remodeling explains the mechanism and importance of histone acetylation using histone acetyl transferase and histone deacetylase enzymes. Histone acetylation is a critical post translational modification of histone proteins that plays a key role in regulating gene expression. it occurs when ace. In this video, we explain major histone modifications including acetylation, methylation, phosphorylation, and ubiquitination, and how they control transcriptional activation and repression.
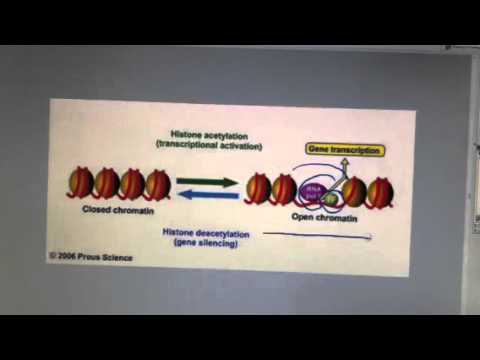
Histone Acetylation Youtube Histone acetylation is a critical post translational modification of histone proteins that plays a key role in regulating gene expression. it occurs when ace. In this video, we explain major histone modifications including acetylation, methylation, phosphorylation, and ubiquitination, and how they control transcriptional activation and repression. Acetylation of histones neutralizes their positive charge, reducing their affinity for negatively charged dna. In this video, we explain histone acetylation – a chemical modification where acetyl groups are added to histone proteins. When it was first discovered, it was thought that acetylation of lysine neutralizes the positive charge normally present, thus reducing affinity between histone and (negatively charged) dna,. Although histones undergo a variety of acylations, the non acetyl acyl marks are found on the same lysine residues where acetylation occurs and are seemingly regulated by the same histone modifying enzymes that govern acetylation.
Comments are closed.