Graphite Structure Vs Diamond
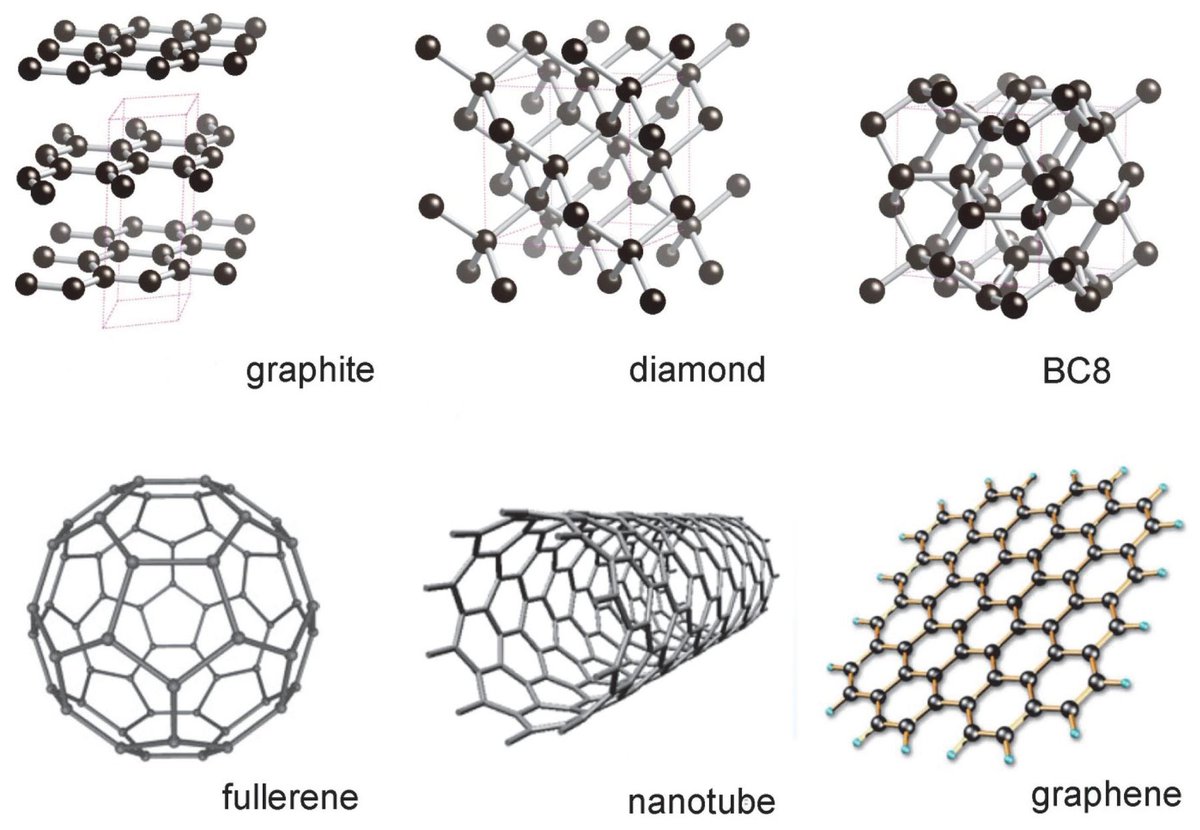
Graphite Structure Vs Diamond Diamond is a type of carbon that has its atoms arranged in a diamond cubic crystal structure. another solid form of carbon known as graphite is the chemically stable form of carbon at ambient temperature and pressure, but diamond almost never transforms to it. Covalent network solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon (iv) oxide). this page relates the structures of covalent network solids to the physical properties of the substances.

Graphite Structure Vs Diamond In diamond, the carbon atoms are arranged in a tetrahedral structure, while in graphite, the carbon atoms are arranged in 2d sheets. the different arrangements of carbon atoms result in different chemical and physical properties for the two allotropes. Learn the key differences between diamond and graphite in chemistry. compare structures, properties, and uses with an easy table for quick revision. Use our revision to learn the structure of diamond and graphite. understand how the structure and bonding influences the properties for igcse chemistry. Diamond is the hardest natural material known, transparent, and electrically insulating. graphite is soft enough to leave marks on paper, opaque black, and conducts electricity. the reason for this dramatic contrast comes down to how the carbon atoms are bonded and arranged at the atomic level.
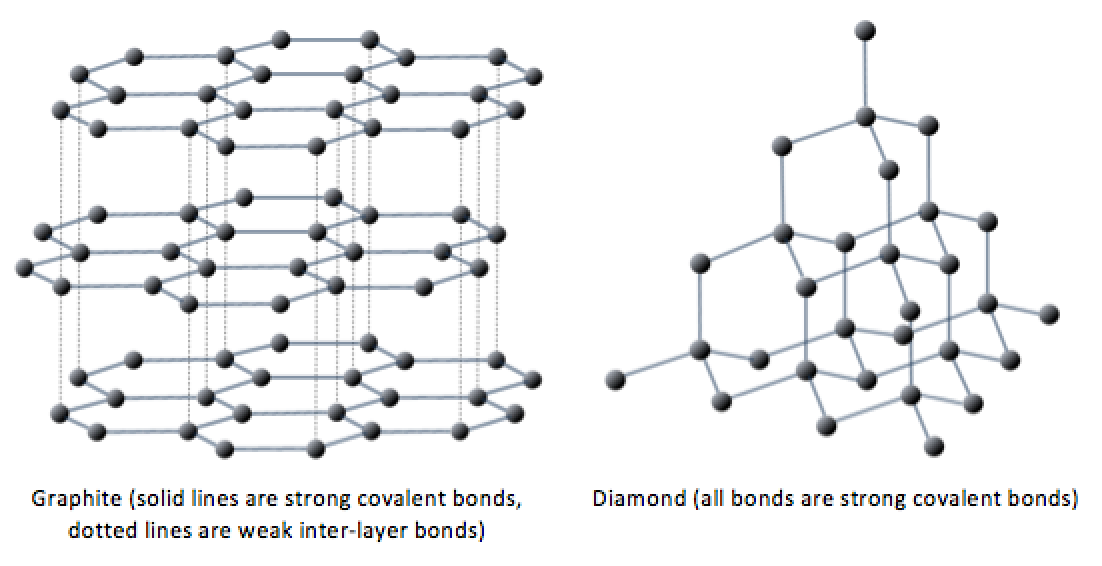
Graphite Structure Vs Diamond Use our revision to learn the structure of diamond and graphite. understand how the structure and bonding influences the properties for igcse chemistry. Diamond is the hardest natural material known, transparent, and electrically insulating. graphite is soft enough to leave marks on paper, opaque black, and conducts electricity. the reason for this dramatic contrast comes down to how the carbon atoms are bonded and arranged at the atomic level. Here is a detailed table comparing and contrasting the properties, structure, and typical applications of diamond and graphite, which are both allotropes of carbon:. Discover how diamond and graphite, both made of pure carbon, exhibit vastly different properties due to their unique atomic structures. Diamond has a covalent network structure and thus does not display intermolecular forces. graphite however has its layers held together by weak van der waals forces. Unlike diamond, graphite utilizes sp2 hybridization, where each carbon atom bonds to only three neighboring atoms. this forms flat, two dimensional sheets arranged in a hexagonal layered structure.
Comments are closed.