Good Lab Practices Rev 3 Pdf

Good Lab Practices Rev 3 Pdf Good lab practices rev 3 peserta mempelajari teknik dasar mikrobiologi seperti preparasi kultur kerja, sterilisasi media, isolasi mikroba, dan penghitungan koloni untuk menghitung jumlah mikroba total secara aerobik.". Through this review, we have made an attempt to explore the uses of glp principles in different fields of science and its acceptability as well as looking for its future perspectives.

Good Laboratory Practices Glp Part 2 Pharmaguideline Pdf Ph The course is designed to provide students with foundational knowledge and practical understanding of standard laboratory practices, procedures, and protocols essential for ensuring safety, reliability, and accuracy in scientific work. The glp regulations set out the rules for good practice and help researchers perform their work in compliance with their own pre established plans and standardized procedures. For most uses, standard precautions (including the use of appropriate laboratory ppe and safe handling practices including working in levs) if they are applied in full, will ensure adequate control. Pedoman praktik laboratorium yang benar depkes 2008.pdf free download as pdf file (.pdf) or read online for free.
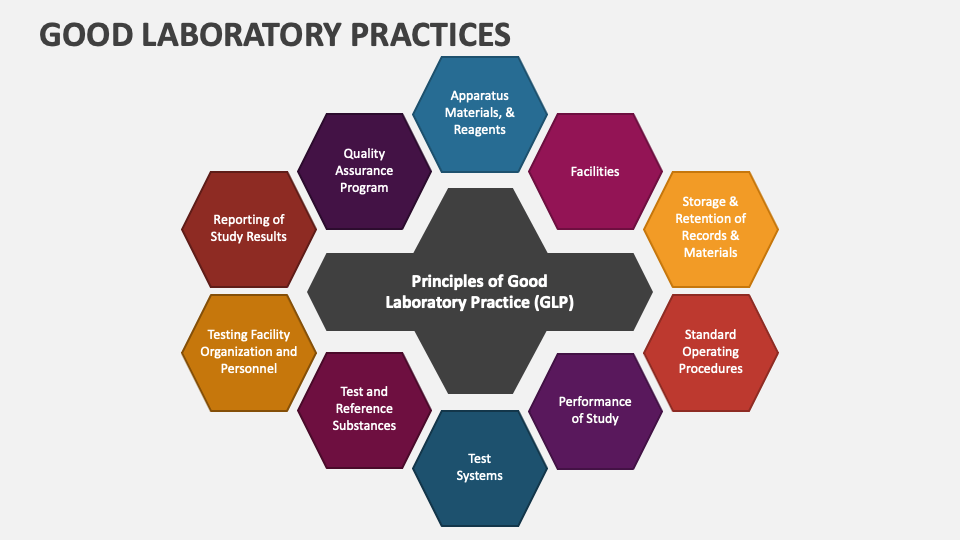
Good Laboratory Practices Powerpoint And Google Slides Template Ppt For most uses, standard precautions (including the use of appropriate laboratory ppe and safe handling practices including working in levs) if they are applied in full, will ensure adequate control. Pedoman praktik laboratorium yang benar depkes 2008.pdf free download as pdf file (.pdf) or read online for free. • good laboratory practices(glp) is a quality system concerned with the organizational process and the conditions under which non clinical health and environmental safety studies are planned, performed, monitored, recorded, archived and reported. Based on the organisation for economic cooperation and development (oecd) principles of glp, the aim of the handbook is to provide laboratories and trainers in disease endemic countries with the necessary technical information for implementing glp programmes. Specifications and performance characteristics of the software described in this manual may be changed at any time without notice. varian reserves the right to make changes in any products herein to improve reliability, function, or design. The document discusses good laboratory practices for microbiological analyses of food. it covers the significance of analysis results, quality control principles, and ensuring the validity and reliability of data.
Comments are closed.