Gas Laws Lab Pdf

Gas Laws Lab Pdf Introduction: in this experiment you will (1) determine whether boyle’s law applies to a mixture of gases (air) and (2) calculate the gas constant, r, by determining the volume of a known amount of gas (h2) at a measured temperature and pressure. Note that in order to use the ideal gas law, the temperature must be in absolute temperature scale units, such as kelvin! in this experiment, you will generate a sample of gas chemically, so that you can determine the number of moles using stoichiometry.

Gas Laws Lab Lab Report For Gas Laws Lab Report Gas Laws Part 1 Regardless of the identity of a gas, all gases exhibit comparable physical behavior. there are simple gas laws that describe the relationship between pairs of the physical properties of gases. This document describes a lab report on the gas laws of boyle's law, charles' law, and gay lussac's law. students used a simulation to change the volume, temperature, and pressure of a gas and observe the effects. An introduction to gas laws in this experiment, the molar mass of butane is determined using the ideal gas law and dalton�. Pv = nrt l gas constant. the value of r is the same for ll ideal gases. an “ideal gas” is defined as one whose behavior can be described exactly by the gas laws presented here. nearly all gases exhibit ideal behavior under stan ard conditions. deviations from ideal behavior are observed, however, for polar gases at low temperatures and.
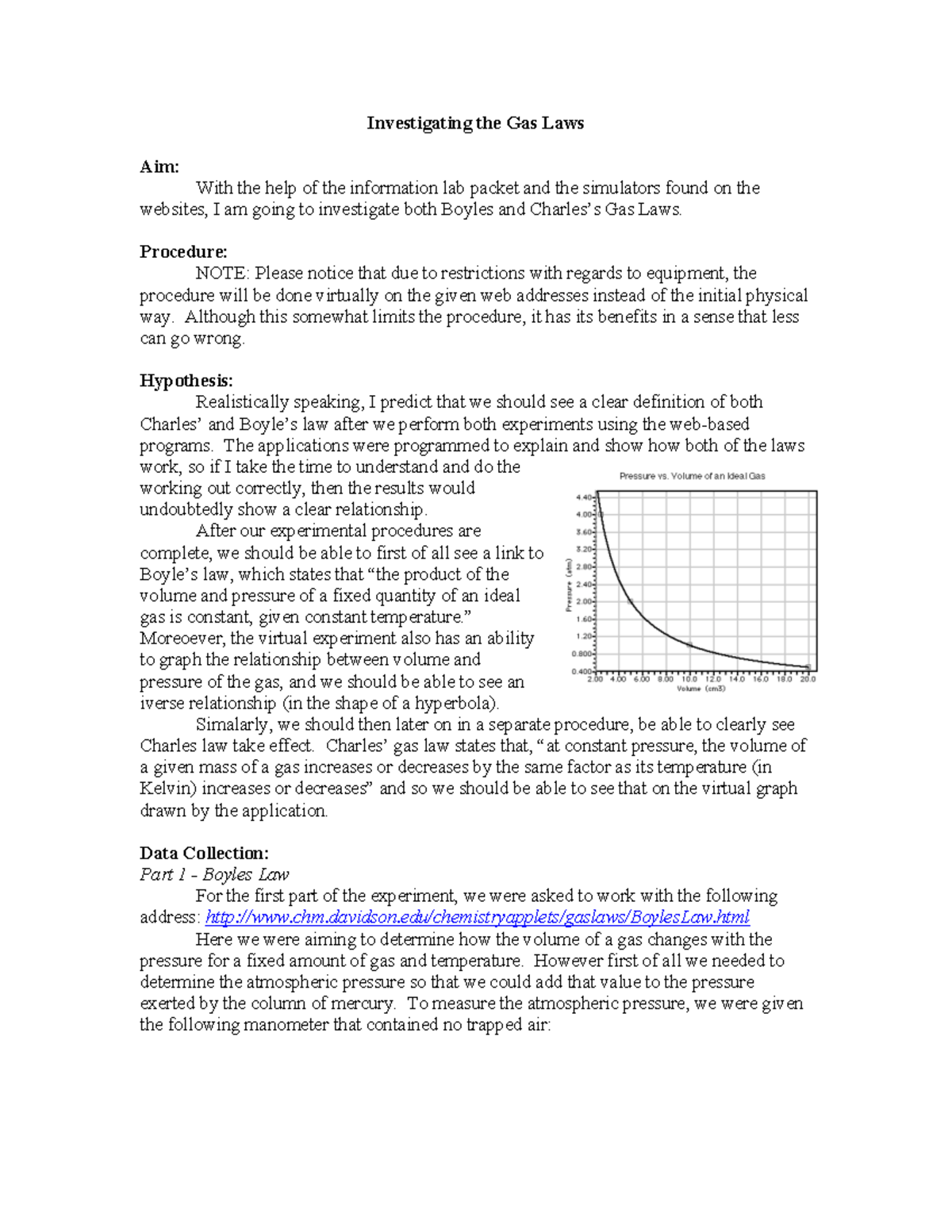
Pdf Gas Laws Lab 1 Lab Investigating The Gas Laws Aim With The An introduction to gas laws in this experiment, the molar mass of butane is determined using the ideal gas law and dalton�. Pv = nrt l gas constant. the value of r is the same for ll ideal gases. an “ideal gas” is defined as one whose behavior can be described exactly by the gas laws presented here. nearly all gases exhibit ideal behavior under stan ard conditions. deviations from ideal behavior are observed, however, for polar gases at low temperatures and. You will investigate this law using a previously recorded data set, including p and v for a gas sample (with n and t constant), by finding the product of p and v for each data point, and comparing these products to see if, in fact, they are equal to each other. This experiment will draw upon your experience with computer based laboratory methods from previous lab activities utilizing the computer. in the first part of the experiment, you will see how changes in volume affect the pressure of a gas. Gay lussac's law: absolute temperature. in other words, if the volume of a gas is held constant, as gas temperature increases, as pressure increases. the correlation between temperature (t) and pressure = the ideal gas law: lussac's law together. it illustrates the relationship between pressure, volume, and temperature for. The purpose of this investigation is to conduct a series of experiments, each of which illustrates a different gas law. you will be given a list of equipment and materials and some general guidelines to help you get started with each experiment.
Comments are closed.