Galvanic Cells Voltaic Cell Definition Working Principle
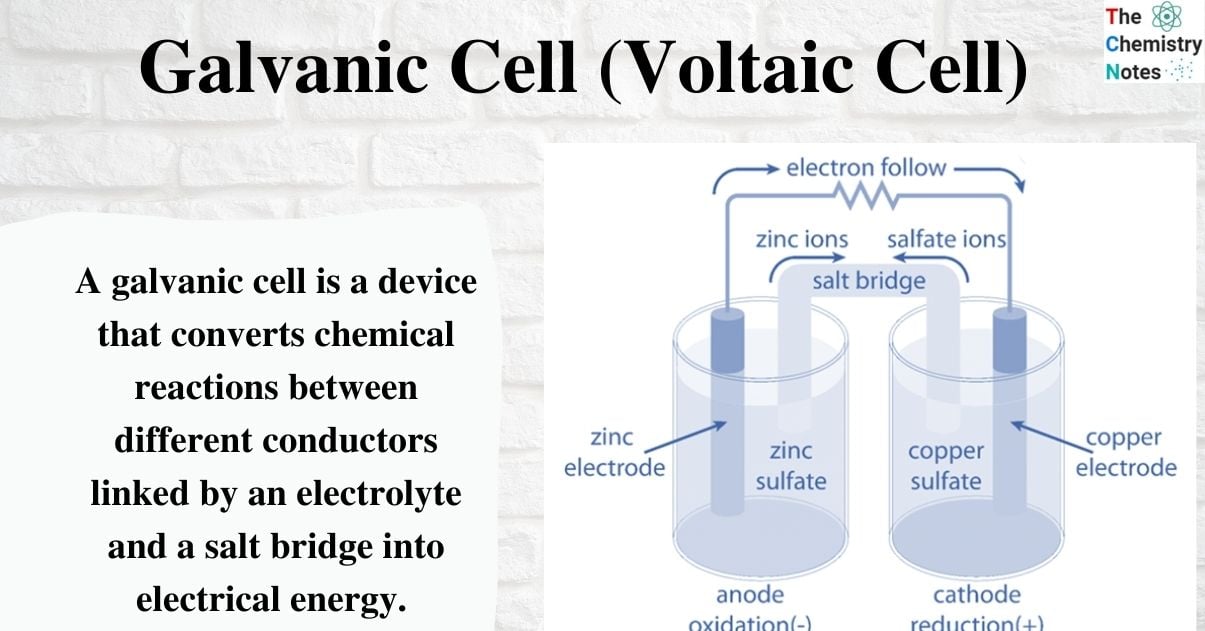
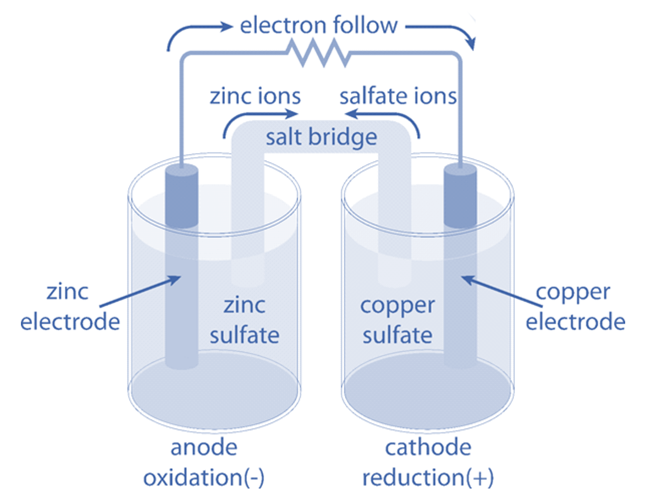
Galvanic Cell Voltaic Cell Definition Working Principle Voltaic or galvanic cells are electrochemical devices that use spontaneous oxidation reduction reactions to generate electricity. an electrochemical cell called a galvanic cell or voltaic cell, respectively named after the scientists luigi galvani and alessandro volta. A galvanic cell, also known as a voltaic cell, is a device that can convert chemical energy into electrical energy through spontaneous redox (oxidation reduction) reactions. it is a type of electrochemical cell that is named after italian scientists luigi galvani and alessandro volta.
Galvanic Cell Voltaic Cell Definition Working Principle Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy. A galvanic cell is an electrochemical cell that converts the chemical energy of spontaneous redox reactions into electrical energy. it is also known as a voltaic cell. In this guide, we explore the structure, working principles, types, and applications of the galvanic cell, ensuring a deep understanding of their functionality. Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy. a battery is a galvanic cell.
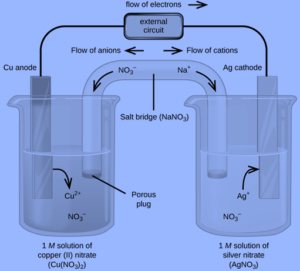
Galvanic Cell Voltaic Cell Definition Working Principle In this guide, we explore the structure, working principles, types, and applications of the galvanic cell, ensuring a deep understanding of their functionality. Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation reduction reactions produce electrical energy. a battery is a galvanic cell. Galvanic cell also called voltaic cell is an electrochemical device that converts spontaneous chemical energy generated in a redox reaction into electrical energy. A galvanic cell uses the energy released from a spontaneous redox reaction to produce an electric current. another common name for galvanic cells is voltaic cells, which is named after another italian physicist, alessandro volta (1745 – 1827), who invented the galvanic (voltaic) cell. A galvanic cell or voltaic cell, named after the scientists luigi galvani and alessandro volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. In lesson 2, we will study a type of electrochemical cell known as a galvanic cell (sometimes referred to as a voltaic cell). a galvanic cell is reliant upon a spontaneous redox reaction.
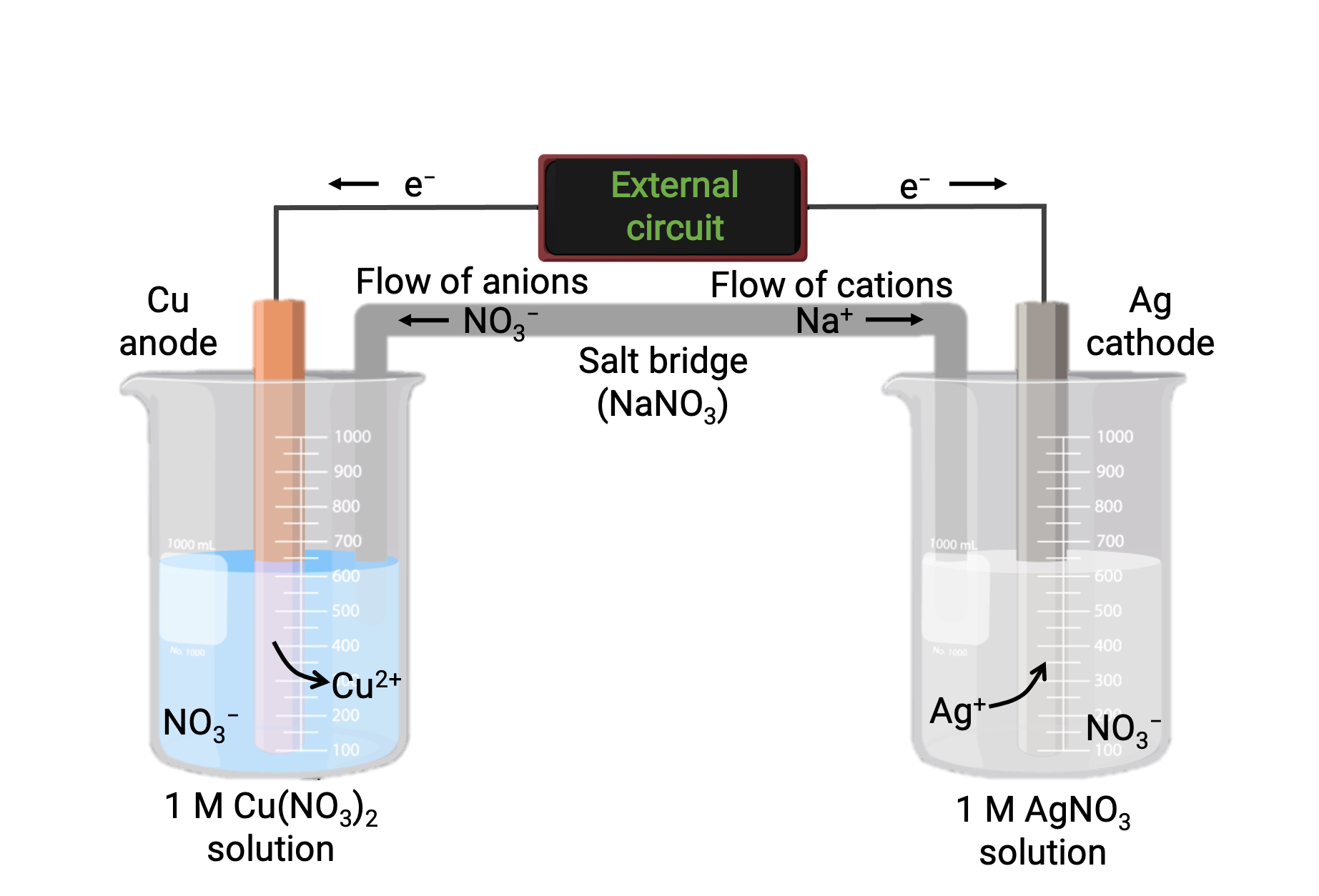
Voltaic Galvanic Cell Galvanic Cells Definition Afcba Galvanic cell also called voltaic cell is an electrochemical device that converts spontaneous chemical energy generated in a redox reaction into electrical energy. A galvanic cell uses the energy released from a spontaneous redox reaction to produce an electric current. another common name for galvanic cells is voltaic cells, which is named after another italian physicist, alessandro volta (1745 – 1827), who invented the galvanic (voltaic) cell. A galvanic cell or voltaic cell, named after the scientists luigi galvani and alessandro volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. In lesson 2, we will study a type of electrochemical cell known as a galvanic cell (sometimes referred to as a voltaic cell). a galvanic cell is reliant upon a spontaneous redox reaction.
Comments are closed.