Free Informed Consent For Research Study Pdf Template %d1%82%d0%b9%d0%b1 Safeinet

Free Informed Consent For Research Study Pdf Template тйб Safeinet He below template for developing an informed consent document to use in your research study is meant to provide structure and guidance to the process, not to serve as your exact informed consent document. Informed consent template free download as word doc (.doc), pdf file (.pdf), text file (.txt) or read online for free. this document provides a template for researchers to use when creating informed consent letters for studies.

Informed Consent Counseling Template Please note that this is a template developed by the who erc to assist the principal investigator in the design of their informed consent forms (icf). it is important that principal investigators adapt their own icfs to the outline and requirements of their particular study. A research informed consent form is used to inform participants in a research study of how the research will be conducted, presented, and reported. the form also confirms the participants' consent to be part of the study, thus freeing the researchers conducting it of any liability. Download a free research consent form template. fill out the form online and save as pdf. customizable and ready to print. New irbmed studies should most often use the latest standard informed consent template available on this webpage. specialty consent templates may be appropriate for low risk studies with simpler study methodologies.

Informed Consent Form Template For Clinical Studies Prntbl Download a free research consent form template. fill out the form online and save as pdf. customizable and ready to print. New irbmed studies should most often use the latest standard informed consent template available on this webpage. specialty consent templates may be appropriate for low risk studies with simpler study methodologies. Include a statement in simple terms to tell people what the research is about. include a statement on how the data will be disseminated (e.g., academic articles, thesis, dissertation, conferences, reports, etc.). The researcher signature section below is not required, but is recommended for research studies involving an in person consent procedure, especially when consent may be obtained by multiple members of the research team. Pending rulemaking for classified human subject research will require additional elements of consent. consent document must be clearly written and understandable to subjects. Download a sample informed consent form for research studies. includes purpose, procedures, risks, confidentiality, and participant rights.

Create Your Informed Consent For Research Study With Our Free Form Include a statement in simple terms to tell people what the research is about. include a statement on how the data will be disseminated (e.g., academic articles, thesis, dissertation, conferences, reports, etc.). The researcher signature section below is not required, but is recommended for research studies involving an in person consent procedure, especially when consent may be obtained by multiple members of the research team. Pending rulemaking for classified human subject research will require additional elements of consent. consent document must be clearly written and understandable to subjects. Download a sample informed consent form for research studies. includes purpose, procedures, risks, confidentiality, and participant rights.

Informed Consent Counseling Template Pending rulemaking for classified human subject research will require additional elements of consent. consent document must be clearly written and understandable to subjects. Download a sample informed consent form for research studies. includes purpose, procedures, risks, confidentiality, and participant rights.
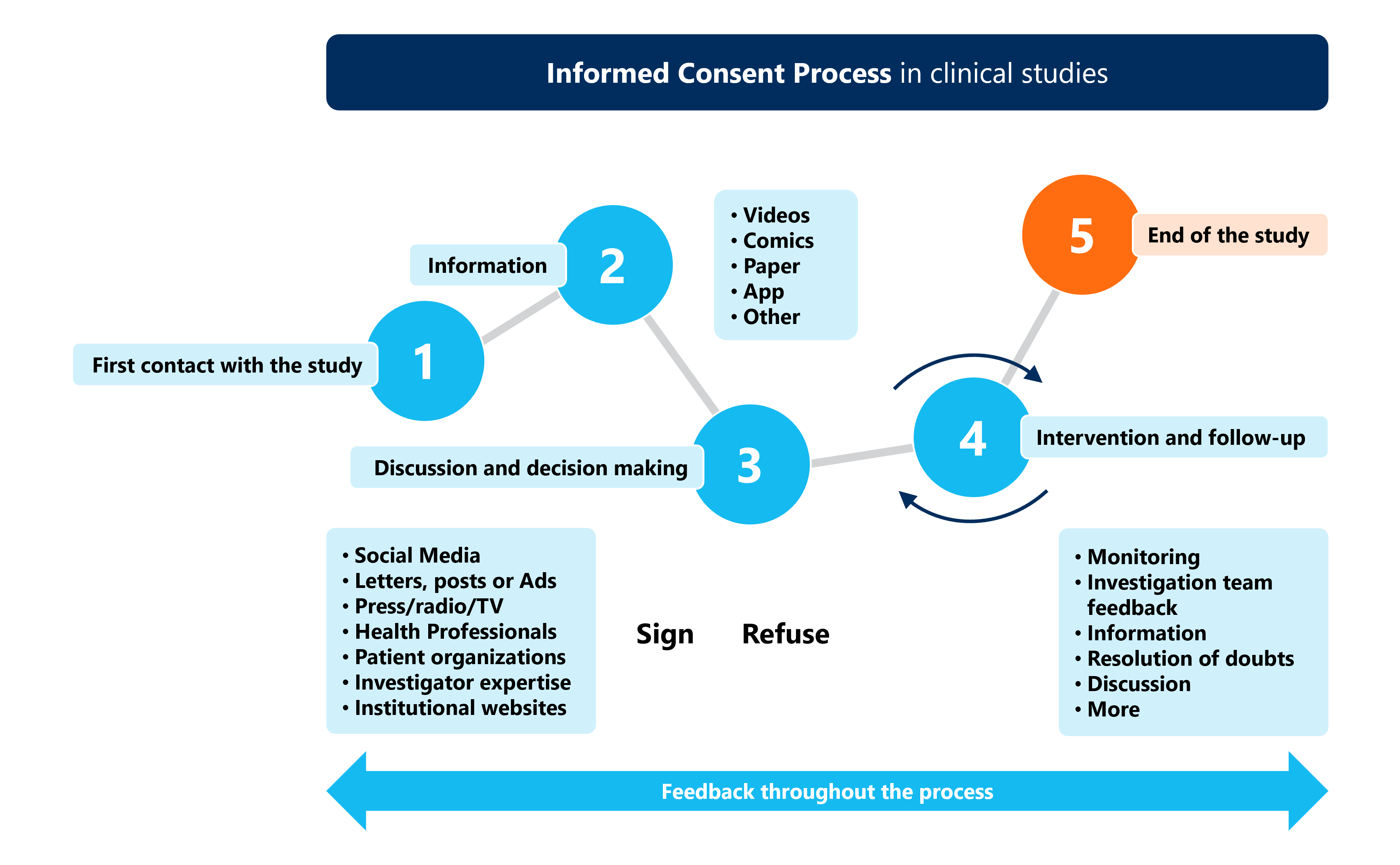
Informed Consent In Clinical Research Ethical Essentials
Comments are closed.