Flowchart For Clinical Trial Phases With Decision Points Research

Flowchart For Clinical Trial Phases With Decision Points Clinical Research Clinical trial flowcharts visually map sequential phases, regulatory checkpoints, patient enrollment milestones, and decision points, enabling researchers and stakeholders to grasp complex timelines at a glance. This slide depicts the process flow of the clinical trial procedure. also, it provides information about mandatory approval points for obtaining necessary information from the regulatory body.
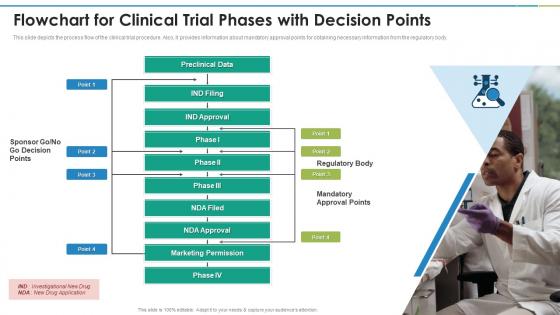
Clinical Trial Phases Flowchart For Clinical Trial Phases With Decision Do you need to generate a flowchart of your study or a systematic review for publication? read here about various ways to generate one. In summary, clinical trials are an essential component of medical research that ensures new treatments are safe and effective before reaching patients. each phase plays a critical role in this process, from initial preclinical research through post market surveillance. A phase 3, multicenter, randomized, open label study to compare the efficacy and safety of golcadomide in combination with rituximab (golca r) vs investigator's choice in participants with relapsed refractory follicular lymphoma. While callaghan innovation is not responsible for the information in this document, if you have a concern or query please contact us on 0800 422 552 or [email protected].
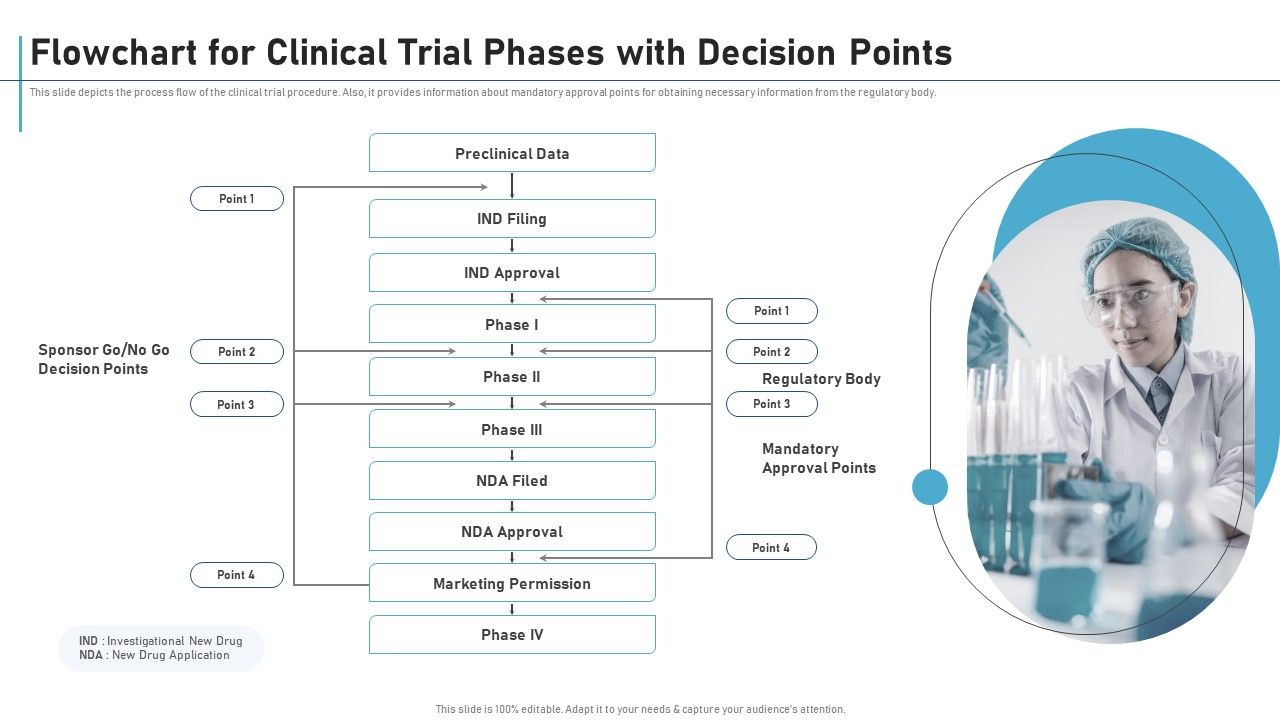
Flowchart For Clinical Trial Phases With Decision Points New Clinical Drug A phase 3, multicenter, randomized, open label study to compare the efficacy and safety of golcadomide in combination with rituximab (golca r) vs investigator's choice in participants with relapsed refractory follicular lymphoma. While callaghan innovation is not responsible for the information in this document, if you have a concern or query please contact us on 0800 422 552 or [email protected]. A clinical study data flow diagram is a visual representation of how data moves within a clinical trial, from its from its capture, review, finalization and archiving. it illustrates the flow of information between stakeholders, systems, and processes involved in the clinical study. Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the. The clinical trial flowcharts are "maps" to help you find clinical trials applicable to various types of cancers. browse our open clinical trials, organized by organ system and modality. This document discusses the clinical trials process from protocol development through study completion. it covers developing the protocol, regulatory documents, patient recruitment, safety reporting, interim reports, and end of study activities.

Clinical Trial Phases Flowchart With Decision Points Ppt Presentation At A clinical study data flow diagram is a visual representation of how data moves within a clinical trial, from its from its capture, review, finalization and archiving. it illustrates the flow of information between stakeholders, systems, and processes involved in the clinical study. Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the. The clinical trial flowcharts are "maps" to help you find clinical trials applicable to various types of cancers. browse our open clinical trials, organized by organ system and modality. This document discusses the clinical trials process from protocol development through study completion. it covers developing the protocol, regulatory documents, patient recruitment, safety reporting, interim reports, and end of study activities.

Process Flow Of Clinical Trial Phases Clinical Research Trial Phases Slides The clinical trial flowcharts are "maps" to help you find clinical trials applicable to various types of cancers. browse our open clinical trials, organized by organ system and modality. This document discusses the clinical trials process from protocol development through study completion. it covers developing the protocol, regulatory documents, patient recruitment, safety reporting, interim reports, and end of study activities.
Comments are closed.