First 10 Alcohols Explained Simply

Alcohols Nomenclature And Classification 44 Off Study with quizlet and memorize flashcards containing terms like methanol, ethanol, propanol and more. An alcohol is an organic compound with a hydroxyl (oh) functional group on an aliphatic carbon atom. because oh is the functional group of all alcohols, we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature.

Introduction To Alcohols Alcohols are named according to the same system as other organic compounds, with the suffix ol used to designate the presence in the molecule of a hydroxyl group. the first step is to consider the number of carbon atoms forming a chain. Alcohols are organic molecules containing the “hydroxyl” functional group, “oh” directly bonded to carbon. the carbon directly attached to oh is technically called the “carbinol” carbon, although this nomenclature is often not introduced in introductory classes. Identify the general structure for an alcohol. identify the structural feature that classifies alcohols as primary, secondary, or tertiary. name alcohols with both common names and iupac names. Learn about alcohols in gcse chemistry: their structure, functional group, naming rules, key uses (fuels, solvents, drinks), and ethanol fermentation.

Alcohols Alcohols Preparation Classification And Uses Of Alcohols Identify the general structure for an alcohol. identify the structural feature that classifies alcohols as primary, secondary, or tertiary. name alcohols with both common names and iupac names. Learn about alcohols in gcse chemistry: their structure, functional group, naming rules, key uses (fuels, solvents, drinks), and ethanol fermentation. For higher chemistry, revise how alcohols are an important class of compounds containing the hydroxyl functional group. Alcohols are covalent molecules; the –oh group in an alcohol molecule is attached to a carbon atom by a covalent bond. we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature. Alcohols are organic compounds that come from crude oil. how to name alcohols and write molecular, condensed formulas, and displayed formulas. Alcohols are hydrocarbons with a hydroxyl (oh) group bonded to a carbon in the chain. the o h bond in alcohols is highly polar, meaning short chain alcohols (methanol and ethanol) are soluble in water. longer chain alcohols are insoluble in water as the carbon chain (alkyl) is not polar.
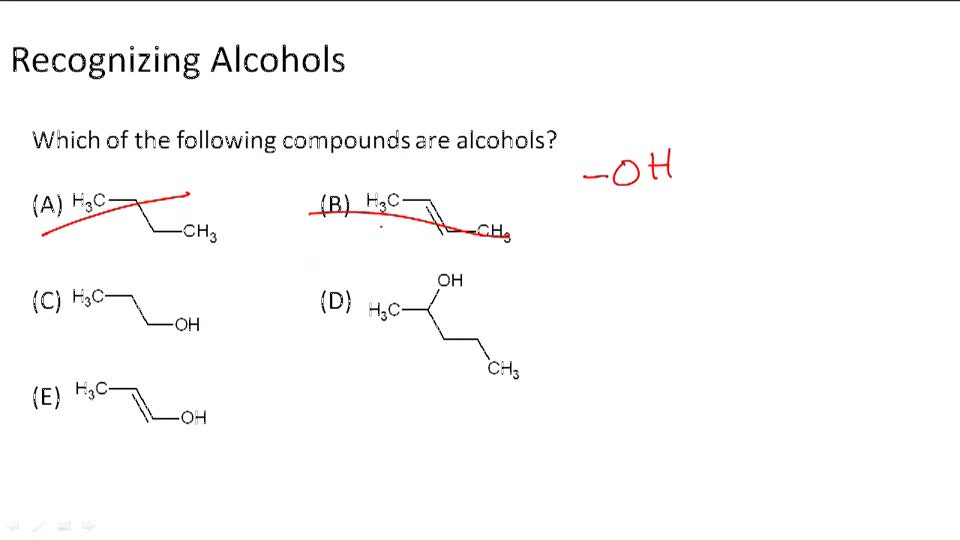
Alcohols Example 1 Video Chemistry Ck 12 Foundation For higher chemistry, revise how alcohols are an important class of compounds containing the hydroxyl functional group. Alcohols are covalent molecules; the –oh group in an alcohol molecule is attached to a carbon atom by a covalent bond. we often represent alcohols by the general formula roh, where r is an alkyl group. alcohols are common in nature. Alcohols are organic compounds that come from crude oil. how to name alcohols and write molecular, condensed formulas, and displayed formulas. Alcohols are hydrocarbons with a hydroxyl (oh) group bonded to a carbon in the chain. the o h bond in alcohols is highly polar, meaning short chain alcohols (methanol and ethanol) are soluble in water. longer chain alcohols are insoluble in water as the carbon chain (alkyl) is not polar.
Comments are closed.