Figure 2 From Progress In Functional Solid Electrolyte Interphases For

Solid Electrolyte Interphases A In Conventional Circumstances The However, the formation of unstable solid electrolyte interphases (seis) and li dendrites, as well as the resultant safety issues initiated by catastrophic dendrite growth, have greatly impeded…. In this review, on the basis of the mechanisms underlying li dendrite formation and growth, strategies for constructing various functional sei films, highlights in structure and property of the.

Solid Electrolyte Interphases A In Conventional Circumstances The In this review, on the basis of the mechanisms underlying li dendrite formation and growth, strategies for constructing various functional sei films, highlights in structure and property of the. In lithium ion batteries, the electrochemical instability of the electrolyte and its ensuing reactive decomposition proceeds at the anode surface within the helmholtz double layer resulting in a buildup of the reductive products, forming the solid electrolyte interphase (sei). Nature abundant sodium metal is regarded as ideal anode material for advanced batteries due to its high specific capacity of 1166 mah g−1 and low redox potential of −2.71 v. however, the uncontrollable dendritic na formation and low coulombic efficiency remain major obstacles to its application. In the production and manufacture of batteries, the sei is naturally formed during the initial charging and discharging process without artificial induction. specifically, the solvent, lithium salts, and additives in the electrolyte are reduced on the lithium anode surface to form an sei in lmbs.
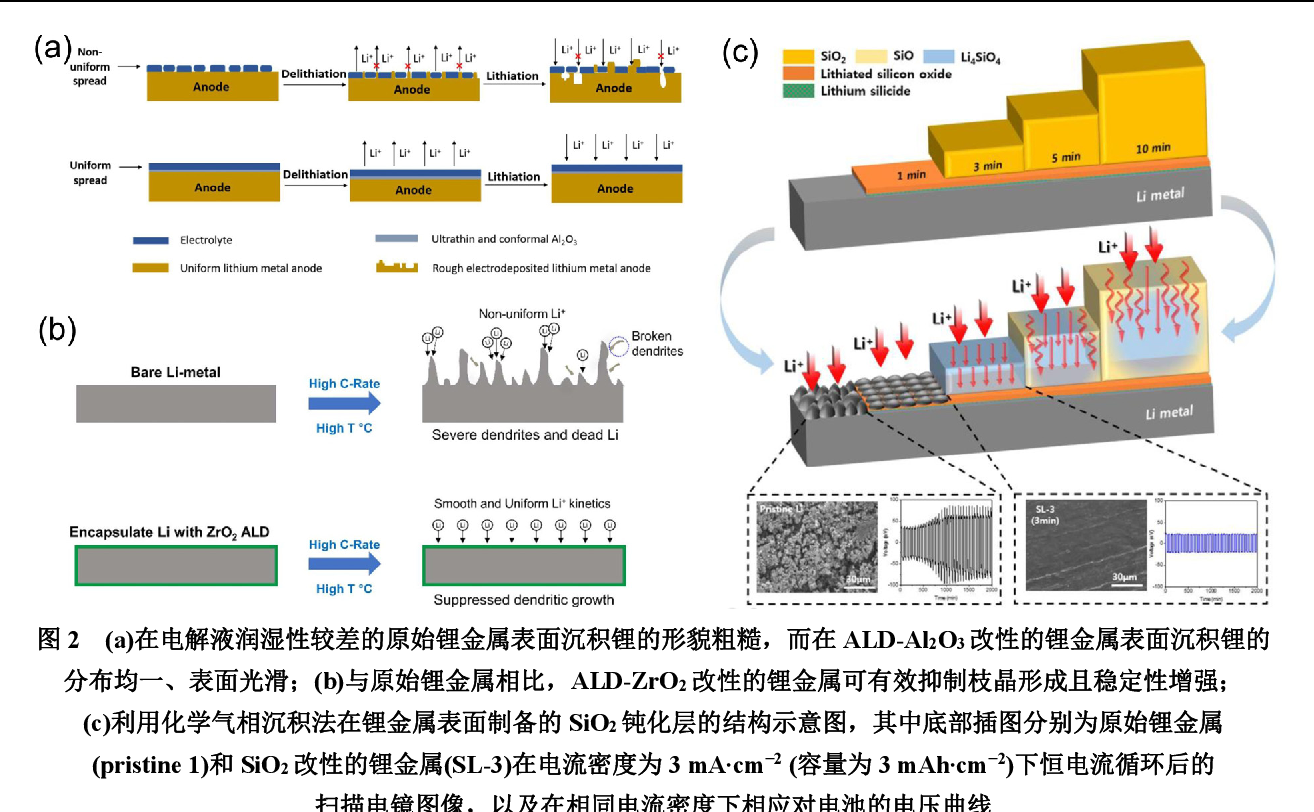
Figure 2 From Progress In Functional Solid Electrolyte Interphases For Nature abundant sodium metal is regarded as ideal anode material for advanced batteries due to its high specific capacity of 1166 mah g−1 and low redox potential of −2.71 v. however, the uncontrollable dendritic na formation and low coulombic efficiency remain major obstacles to its application. In the production and manufacture of batteries, the sei is naturally formed during the initial charging and discharging process without artificial induction. specifically, the solvent, lithium salts, and additives in the electrolyte are reduced on the lithium anode surface to form an sei in lmbs. The formation of a stable solid electrolyte interphase (sei) layer is very important for improving the cycling stability and safety of lithium metal batteries (lmbs). In this review, on the basis of the mechanisms underlying li dendrite formation and growth, strategies for constructing various functional sei films, highlights in structure and property of the films, and their effects on the performance of li metal anodes are summarized. Here, the authors study the formation and long term evolution of the sei near open circuit conditions in symmetric silicon cells containing different electrolyte chemistries. Researchers have explored various approaches to address these issues in oxide ses, encompassing electrolyte modification, the creation of artificial solid state electrolyte interfaces, and composite li anodes.
Comments are closed.