Fda Rejects Psychedelic Mdma Assisted Therapy For Ptsd

Fda Rejects Mdma Assisted Therapy For Ptsd Medpage Today Mdma assisted psychotherapy to treat ptsd became a front runner for approval in 2017, when the nonprofit multidisciplinary association for psychedelic studies (maps) won fda’s “breakthrough therapy” designation for the treatment based on data from phase 2 trials. Maps, the multidisciplinary association for psychedelic studies, while not working directly with the fda, held a press briefing to further discuss the contents of the crl on rejection of mdma assisted therapy for ptsd.

Fda Panel Rejects Mdma Assisted Therapies For Ptsd In Veterans Fox News Food and drug administration issues complete response letter rejecting lykos therapeutics' mdma assisted therapy for ptsd, demanding additional clinical trials despite promising efficacy data. San jose, ca; september 4, 2025 — in august 2024, the u.s. food and drug administration (fda) issued a complete response letter (crl) to lykos therapeutics, declining to approve mdma assisted therapy for posttraumatic stress disorder (ptsd). Washington (ap) — federal health regulators on friday declined to approve the psychedelic drug mdma as a therapy for ptsd, a major setback for groups seeking a breakthrough decision in favor of using mind altering substances to treat serious mental health conditions. The fda rejected lykos therapeutics’ application to use mdma for ptsd treatment, citing the need for further safety and efficacy studies.

Fda Panel Rejects Mdma For Ptsd Amid Safety And Methodology Concerns In Washington (ap) — federal health regulators on friday declined to approve the psychedelic drug mdma as a therapy for ptsd, a major setback for groups seeking a breakthrough decision in favor of using mind altering substances to treat serious mental health conditions. The fda rejected lykos therapeutics’ application to use mdma for ptsd treatment, citing the need for further safety and efficacy studies. This article will dive deeper into the fda’s rejection of mdma to understand what went wrong in this landmark effort toward an fda approval of a psychedelic medicine, which is the first to have reached this critical juncture. The food and drug administration's long awaited complete response letter rejecting mdma assisted therapy for post traumatic stress disorder has finally been released, revealing specific concerns that extend beyond the safety and efficacy data that dominated public discourse—and triggering a fundamental restructuring of the most prominent company in psychedelic medicine. One year after fda’s rejection of lykos therapeutics’ mdma assisted therapy for ptsd application, insiders and observers reflect on what went wrong and what comes next. Drugmaker lykos therapeutics announced on aug. 9, 2024, that the food and drug administration declined to approve the company’s application for the use of mdma assisted therapy in the.
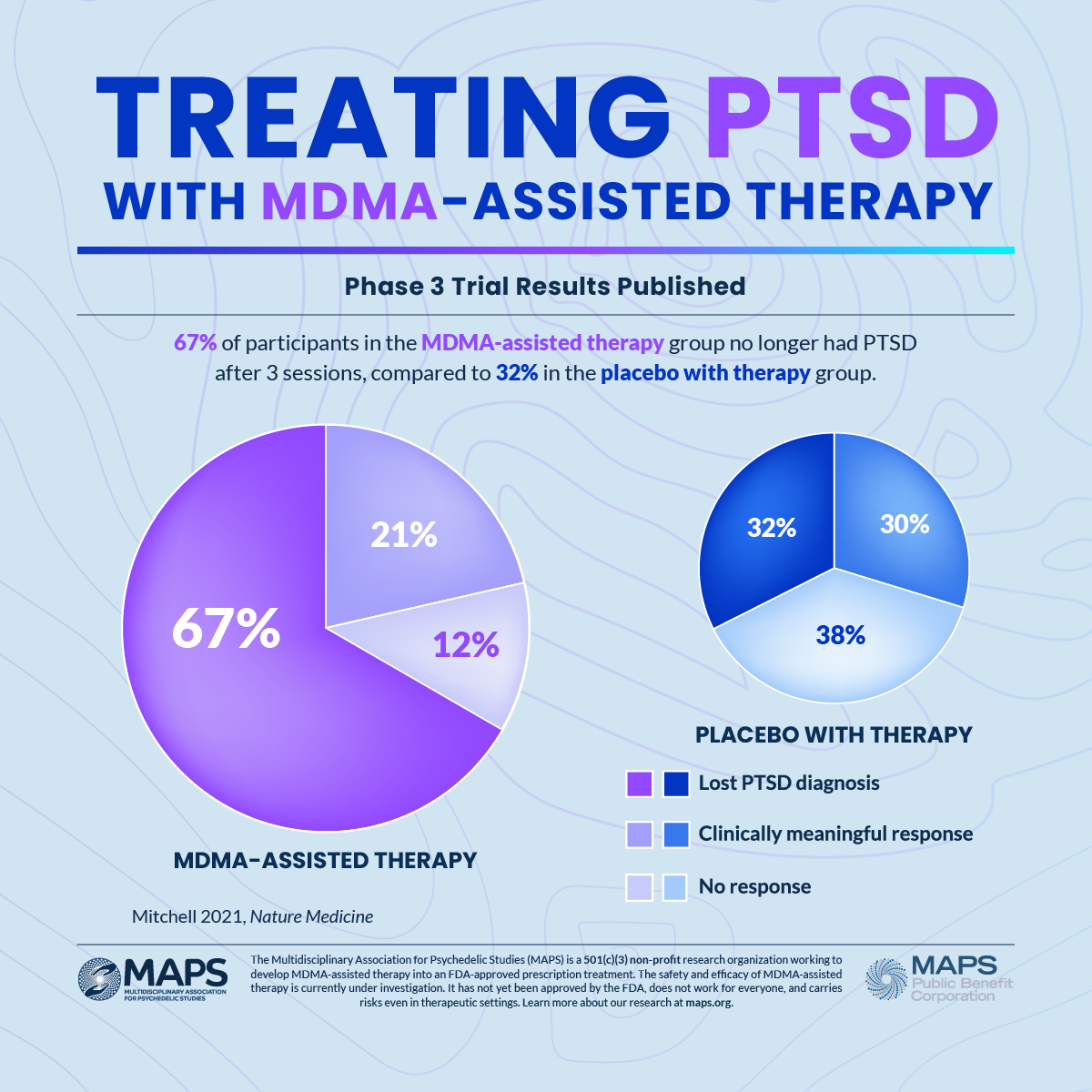
Maps Phase 3 Trial Of Mdma Assisted Therapy For Ptsd Achieves This article will dive deeper into the fda’s rejection of mdma to understand what went wrong in this landmark effort toward an fda approval of a psychedelic medicine, which is the first to have reached this critical juncture. The food and drug administration's long awaited complete response letter rejecting mdma assisted therapy for post traumatic stress disorder has finally been released, revealing specific concerns that extend beyond the safety and efficacy data that dominated public discourse—and triggering a fundamental restructuring of the most prominent company in psychedelic medicine. One year after fda’s rejection of lykos therapeutics’ mdma assisted therapy for ptsd application, insiders and observers reflect on what went wrong and what comes next. Drugmaker lykos therapeutics announced on aug. 9, 2024, that the food and drug administration declined to approve the company’s application for the use of mdma assisted therapy in the.

Psychedelic Assisted Therapy Mini Series Part 1 An Overview Of Mdma One year after fda’s rejection of lykos therapeutics’ mdma assisted therapy for ptsd application, insiders and observers reflect on what went wrong and what comes next. Drugmaker lykos therapeutics announced on aug. 9, 2024, that the food and drug administration declined to approve the company’s application for the use of mdma assisted therapy in the.
Comments are closed.