Fda Orange Book

The Fda Orange Book Special Edition Pdf Generic Drug Food And The orange book lists drug products approved by fda and their patent and exclusivity information. search the database by various criteria or access the publications, supplements, appendices, and guidance documents. The approved drug products with therapeutic equivalence (orange book or ob) is a list of drugs approved under section 505 of the federal food, drug and cosmetic act and provides consumers timely updates on these products.
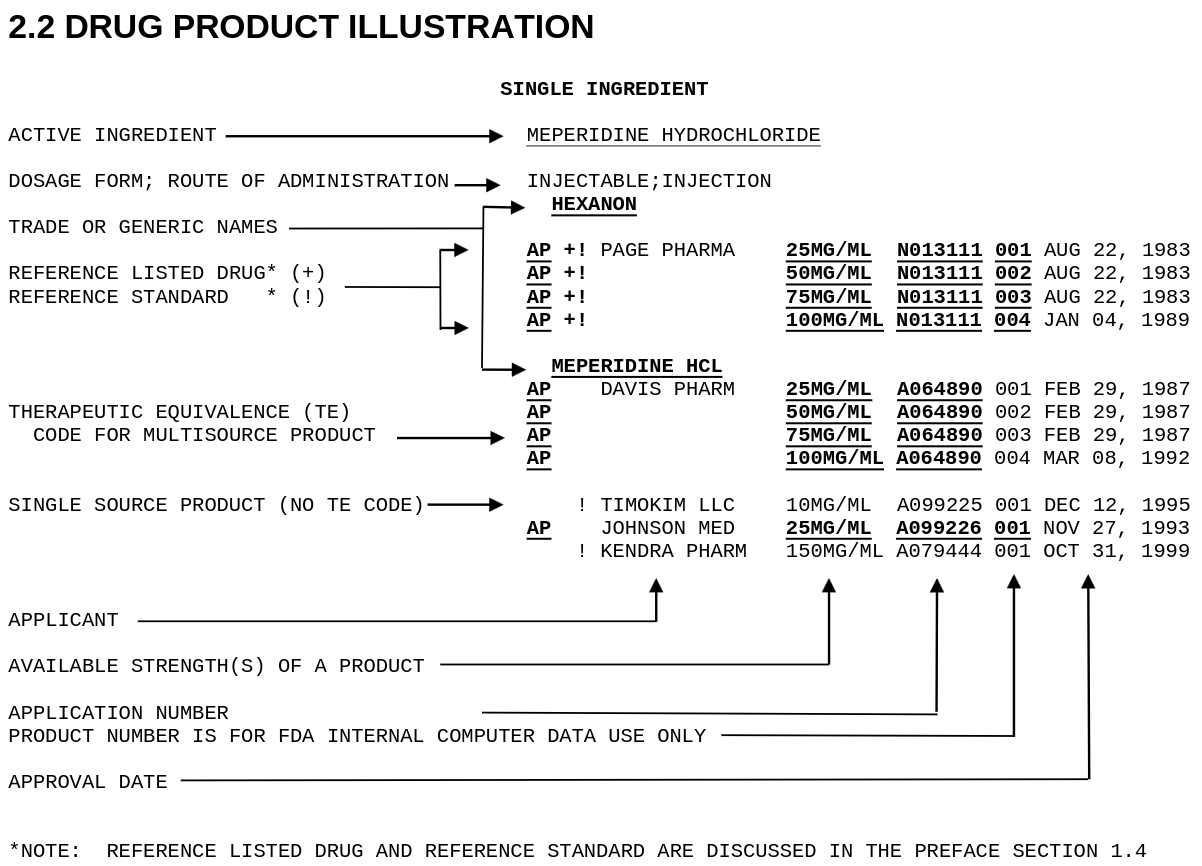
Fda Orange Book Rxdatalab The orange book "identifies drug products approved on the basis of safety and effectiveness by the food and drug administration (fda) under the federal food, drug, and cosmetic act (the act). Download pdfs of the fda orange book from 1980 to 2025, including annual editions and monthly supplements. the fda orange book lists approved drug products with therapeutic equivalence evaluations. Download compressed data files for the orange book, a database of approved drug products and patents in the u.s. see the field descriptions for products, patents, and exclusivity information. Officially titled approved drug products with therapeutic equivalence evaluations, the orange book is published by the united states fda. it provides a comprehensive list of all drug products approved based on safety and efficacy.

Fda Orange Book Rxdatalab Download compressed data files for the orange book, a database of approved drug products and patents in the u.s. see the field descriptions for products, patents, and exclusivity information. Officially titled approved drug products with therapeutic equivalence evaluations, the orange book is published by the united states fda. it provides a comprehensive list of all drug products approved based on safety and efficacy. Although some outside users repackage the information, the only definitive source for therapeutic equivalence (te) and brand name (“innovator”) drug data, as well as patent and exclusivity data, is the orange book. The orange book lists drug products approved by the fda under the federal food, drug, and cosmetic act, along with their therapeutic equivalence evaluations and patents. it is a public information source for drug selection and health care costs, but not an official fda action. Federal government websites often end in .gov or .mil. before sharing sensitive information, make sure you're on a federal government site. Chemical names were collected from the ingredients listed in the product.txt file contained within the zipfile available on the "orange book data files (compressed)" link available on this page.
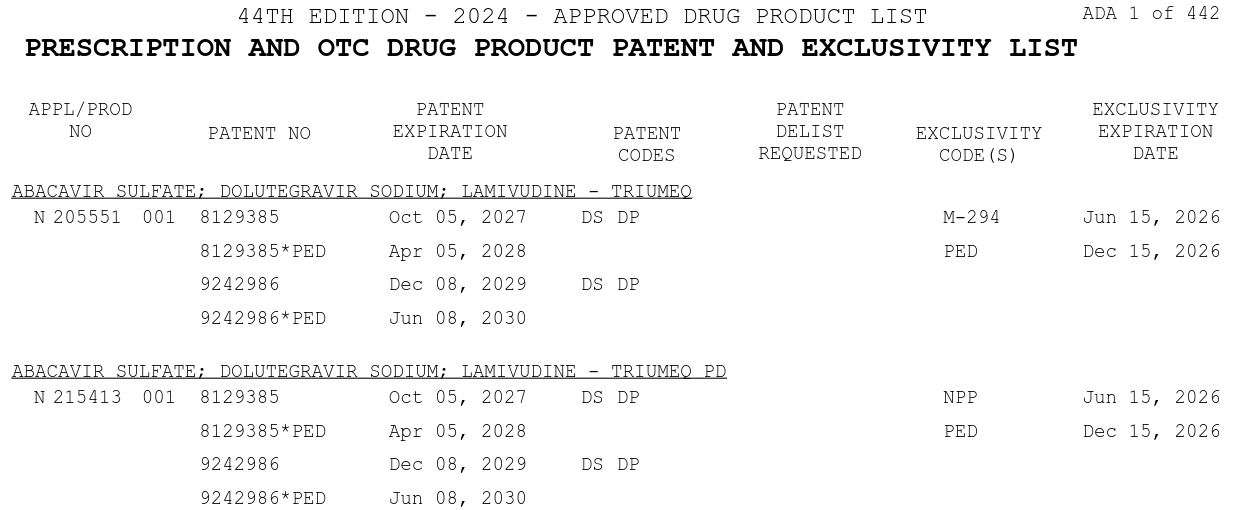
Fda Orange Book Rxdatalab Although some outside users repackage the information, the only definitive source for therapeutic equivalence (te) and brand name (“innovator”) drug data, as well as patent and exclusivity data, is the orange book. The orange book lists drug products approved by the fda under the federal food, drug, and cosmetic act, along with their therapeutic equivalence evaluations and patents. it is a public information source for drug selection and health care costs, but not an official fda action. Federal government websites often end in .gov or .mil. before sharing sensitive information, make sure you're on a federal government site. Chemical names were collected from the ingredients listed in the product.txt file contained within the zipfile available on the "orange book data files (compressed)" link available on this page.

Fda Orange Book Archives Biotechblog Federal government websites often end in .gov or .mil. before sharing sensitive information, make sure you're on a federal government site. Chemical names were collected from the ingredients listed in the product.txt file contained within the zipfile available on the "orange book data files (compressed)" link available on this page.

Pdf Orange Book By Fda Approved Drug Products 41st Edition 2021
Comments are closed.