Fda Medical Device Reporting

Adverse Event Reporting For Medical Devices Complete Fda Medical Requirements for reporting medical device problems, including malfunctions and adverse events (serious injuries or deaths) associated with medical devices. Learn about the requirements for reporting deaths, serious injuries, and device malfunctions for device user facilities, manufacturers, importers, and distributors. find out the terms and conditions for adverse event reporting, followup, and record keeping.
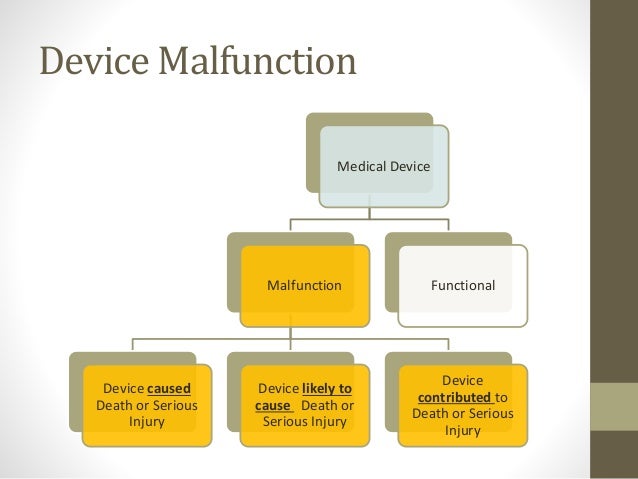
Fda And Medical Device Reporting Patients, healthcare professionals and consumers who find a problem related to a medical device are encouraged to report medical device adverse events or product problems to the fda through medwatch, the fda safety information and adverse event reporting program. User facilities report when they become aware of information that reasonably suggests that a device may have caused or contributed to a patient death or serious injury. Ensure compliance and safety with our medical device reporting guide. learn how medical device reporting impacts manufacturers and healthcare facilities. Medical device reporting (mdr) regulations require manufacturers to report to fda device related incidents, deaths, serious injuries, and device malfunctions which are likely to cause or contribute to death or serious injury if they were to occur.

4 Fda Medical Device Reporting Procedures For Life Science Companies Ensure compliance and safety with our medical device reporting guide. learn how medical device reporting impacts manufacturers and healthcare facilities. Medical device reporting (mdr) regulations require manufacturers to report to fda device related incidents, deaths, serious injuries, and device malfunctions which are likely to cause or contribute to death or serious injury if they were to occur. Every time you report a side effect, device malfunction, or product quality problem to the fda, you’re contributing to a vast early warning system that protects public health. Lii electronic code of federal regulations (e cfr) title 21—food and drugs chapter i—food and drug administration, department of health and human services subchapter h—medical devices part 803—medical device reporting. Learn the essentials of medical device adverse event reporting with our comprehensive fda mdr guide 2025. ensure compliance and avoid penalties. This article will review the medical device reporting (mdr) requirements, important stakeholders, the process required for reporting a medical device problem as a medical device manufacturer or user, and the critical deadlines for medical device reports.

Fda Medical Device Reporting Every time you report a side effect, device malfunction, or product quality problem to the fda, you’re contributing to a vast early warning system that protects public health. Lii electronic code of federal regulations (e cfr) title 21—food and drugs chapter i—food and drug administration, department of health and human services subchapter h—medical devices part 803—medical device reporting. Learn the essentials of medical device adverse event reporting with our comprehensive fda mdr guide 2025. ensure compliance and avoid penalties. This article will review the medical device reporting (mdr) requirements, important stakeholders, the process required for reporting a medical device problem as a medical device manufacturer or user, and the critical deadlines for medical device reports.
Comments are closed.