Fda Medical Device Classification Reghelps Src
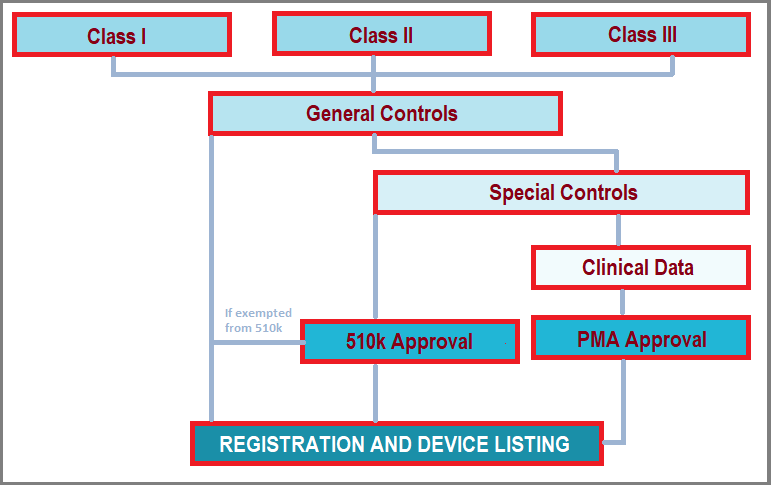
Fda Medical Device Classification Reghelps Src The us food and drug administration (fda) classified medical devices depending on the risk they pose to the user. the fda divides devices into three classes: class i (low risk), class ii (moderate risk), and class iii (high risk) (high risk). To find the classification of your device, as well as whether any exemptions may exist, you need to find the regulation number that is the classification regulation for your device.

Medical Device Manufacturing Supplementing the general food and drug administration procedures governing advisory committees (part 14 of this chapter), this part also provides procedures for manufacturers, importers, and other interested persons to participate in proceedings to classify and reclassify devices. Complete guide to fda device classes i, ii & iii. learn classification criteria, regulatory pathways, examples & requirements for 2025. Medical devices are classified both by medical specialty (i.e., classification panels) and by the risk posed to the consumer (i.e., regulatory classes). In 1976, the medical device amendments were added to the united states (u.s.) federal food, drug and cosmetic act (fd&c act) of 1938, establishing the current risk based classification system for all medical devices the fda regulates.
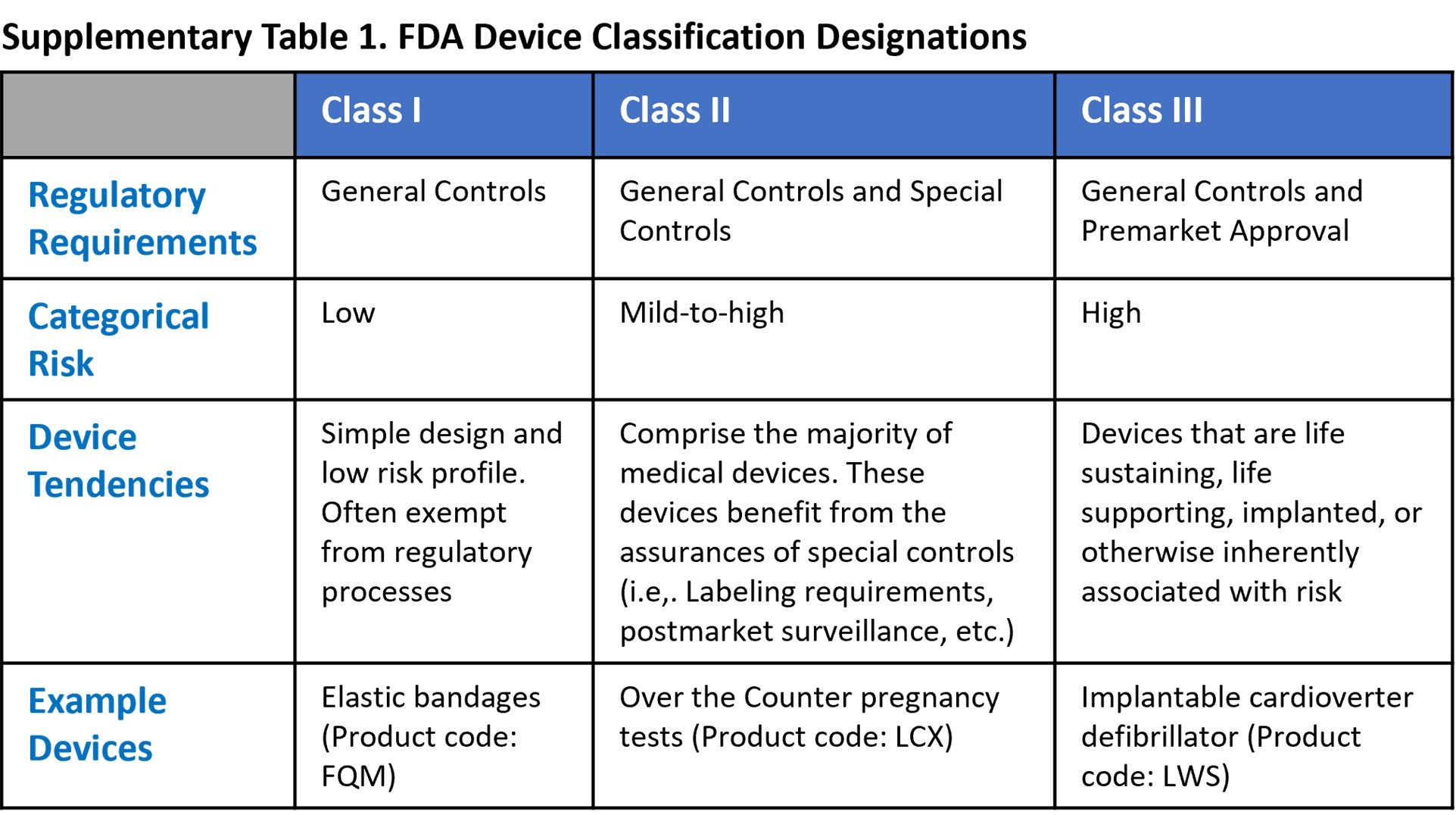
Fda Classification System Medical devices are classified both by medical specialty (i.e., classification panels) and by the risk posed to the consumer (i.e., regulatory classes). In 1976, the medical device amendments were added to the united states (u.s.) federal food, drug and cosmetic act (fd&c act) of 1938, establishing the current risk based classification system for all medical devices the fda regulates. For six preamendments, unclassified device types, you will be asked to provide input to fda on the classification for each one: class iii, class ii, or class i. We combine technical precision with regulatory mastery to guide your medical device from concept to market—ensuring safety, effectiveness, and patient trust every step of the way. This database contains device names and their associated product codes. the name and product code identify the generic category of a device for fda. U.s. food and drug administration.

Medical Devices Angela N Johnson For six preamendments, unclassified device types, you will be asked to provide input to fda on the classification for each one: class iii, class ii, or class i. We combine technical precision with regulatory mastery to guide your medical device from concept to market—ensuring safety, effectiveness, and patient trust every step of the way. This database contains device names and their associated product codes. the name and product code identify the generic category of a device for fda. U.s. food and drug administration.

Medical Device Fda Regulations Infographic Synectic This database contains device names and their associated product codes. the name and product code identify the generic category of a device for fda. U.s. food and drug administration.
Comments are closed.