Fda Issues Complete Response Letter For Rpi In Advanced Melanoma
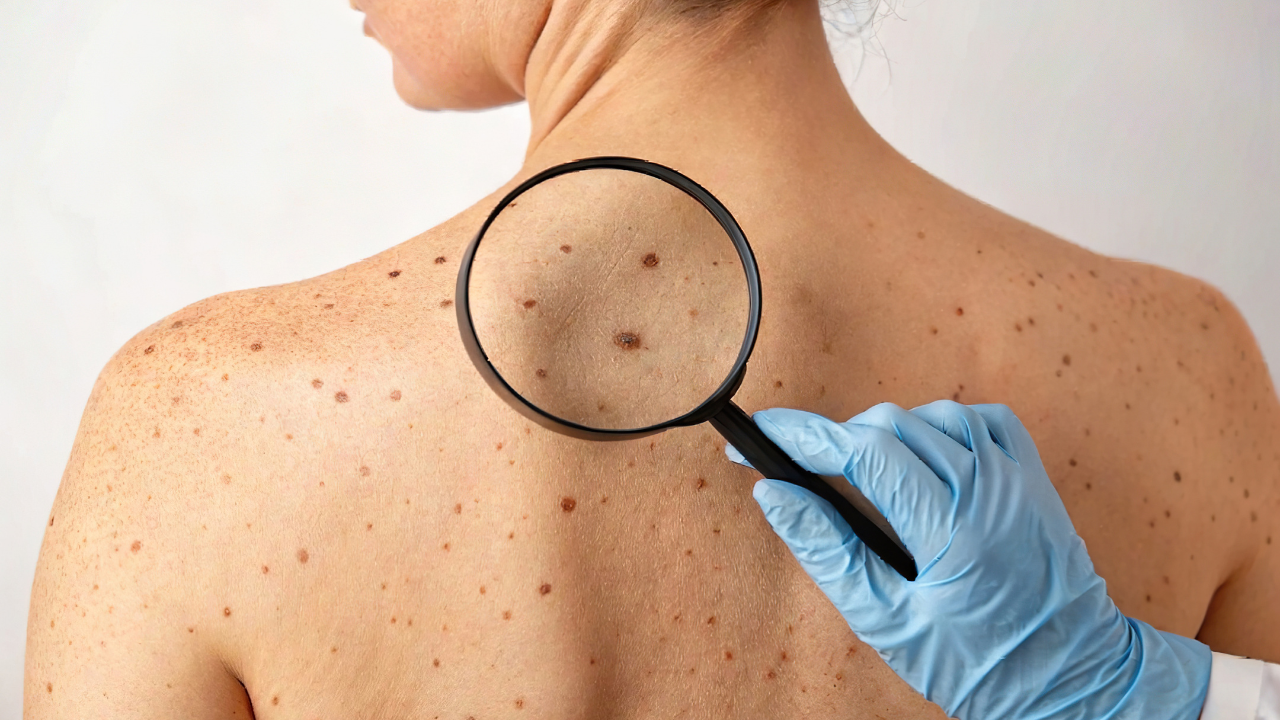
Fda Issues Complete Response Letter To Replimune For Rp1 Combination The fda has issued a complete response letter (crl) to vusolimogene oderparepvec (rp1) in combination with nivolumab (opdivo) for the treatment of patients with advanced melanoma who were previously administered an anti–pd 1 regimen, according to a news release from the drug’s developer, replimune. 1. The company requested feedback, however, the fda did not respond and subsequently accepted the resubmission as a complete response to the july 2025 crl. the fda raised several points related to tumor assessment methodology.
Fda Issues Complete Response Letter To Replimune For Rp1 Combination The fda has issued a complete response letter (crl) to replimune group regarding its biologics license application (bla) for rp1 (vusolimogene oderparepvec) in combination with nivolumab for the treatment of advanced melanoma. We have completed a comprehensive review of all the submissions relating to this bla for vusolimogene oderparepvec in combination with nivolumab for adult patients with unresectable advanced cutaneous melanoma who experienced disease progression with a programmed death receptor 1 (pd 1) blocking antibody based therapy. we performed a reanalysis of data from study rpl 001 16 along with the. The fda has issued a complete response letter (crl) for a biologics license application (bla) seeking the approval of vusolimogene oderparepvec (rp1) in combination with nivolumab. The us food and drug administration (fda) has issued a complete response letter (crl) to replimune group’s biologics licence application (bla) concerning the rp1 therapy in combination with nivolumab for advanced melanoma.
.png)
Fda Issues Complete Response Letter To Replimune For Rp1 Combination The fda has issued a complete response letter (crl) for a biologics license application (bla) seeking the approval of vusolimogene oderparepvec (rp1) in combination with nivolumab. The us food and drug administration (fda) has issued a complete response letter (crl) to replimune group’s biologics licence application (bla) concerning the rp1 therapy in combination with nivolumab for advanced melanoma. Replimune group inc. today announced that it has received a complete response letter (crl) from the fda regarding its biologics license application (bla) for rp1 (vusolimogene oderparepvec), an oncolytic immunotherapy, in combination with nivolumab (opdivo) for the treatment of advanced melanoma. 1. The fda has issued a complete response letter to replimune indicating it is unable to approve the biologics license application for vusolimogene oderparepvec in combination with nivolumab. Replimune receives complete response letter from the fda for rp1 biologics license application for the treatment of advanced melanoma. Today, the us fda issued a complete response letter (crl) to replimune group regarding its biologics license application (bla) for rp1 (vusolimogene oderparepvec) in combination with nivolumab.
Comments are closed.