Fda Expands Approval Of Imfinzi To Reduce The Risk Of Nsclc Progression

Us Fda Approves Imfinzi For Unresectable Stage Iii Non Small Cell Lung The approval by the food and drug administration (fda) was based on positive results from the pivotal aegean trial, which were published in the new england journal of medicine in october 2023. The addition of perioperative imfinzi to neoadjuvant chemotherapy improves pathological complete response (pcr) rates and prolongs cancer free survival in patients with stage ii to iiib nsclc, according to findings from the phase 3 aegean clinical trial.

Us Fda Approves Imfinzi For Unresectable Stage Iii Non Small Cell Lung The approval of imfinzi for the treatment of stage iii, unresectable nsclc was based on a randomized trial of 713 patients whose cancer had not progressed after completing chemotherapy and. On august 15, 2024, the us food and drug administration (fda) approved a new treatment regimen for people newly diagnosed with early stage, resectable (able to be removed by surgery) non small cell lung cancer (nsclc) whose tumors do not have known egfr mutations or alk rearrangements. The u.s. food and drug administration (fda) recently approved the use of the monoclonal antibody durvalumab (imfinzi) in combination with chemotherapy for treatment of adults with resectable, early stage (iia–iiib) non–small cell lung cancer (nsclc) and no known egfr mutations or alk rearrangements. The us food and drug administration has approved imfinzi (durvalumab) for the treatment of patients with stage iii non small cell lung cancer (nsclc) whose tumours are not able to be surgically removed (unresectable) and whose cancer has not progressed after treatment with chemotherapy and radiation (chemoradiation).

Us Fda Approves Imfinzi For Unresectable Stage Iii Non Small Cell Lung The u.s. food and drug administration (fda) recently approved the use of the monoclonal antibody durvalumab (imfinzi) in combination with chemotherapy for treatment of adults with resectable, early stage (iia–iiib) non–small cell lung cancer (nsclc) and no known egfr mutations or alk rearrangements. The us food and drug administration has approved imfinzi (durvalumab) for the treatment of patients with stage iii non small cell lung cancer (nsclc) whose tumours are not able to be surgically removed (unresectable) and whose cancer has not progressed after treatment with chemotherapy and radiation (chemoradiation). Fda’s latest approval of imfinzi (durvalumab) in combination with chemotherapy for treating resectable non small cell lung cancer before and after surgery is based on phase iii trial results showing that the regimen reduced the risk of recurrence, progression, or death by 32%. Durvalumab (imfinzi; astratzeneca) in combination with chemotherapy has been approved by the fda for the treatment of adult patients with resectable early (iia iiib) non small cell lung cancer (nsclc) and no known epidermal growth factor receptor (egfr) mutations or anaplastic lymphoma kinase (alk) rearrangements. The approval by the food and drug administration (fda) was based on positive results from the pivotal aegean trial, which were published in the new england journal of medicine in october 2023. Astrazeneca is backing imfinzi’s small cell lung cancer fda bid with data from the phase iii adriatic study, which showed that imfinzi could lower the risk of death by 27% in these patients as compared with placebo. the pd l1 blocker also reduced the risk of disease progression or death by 24%.
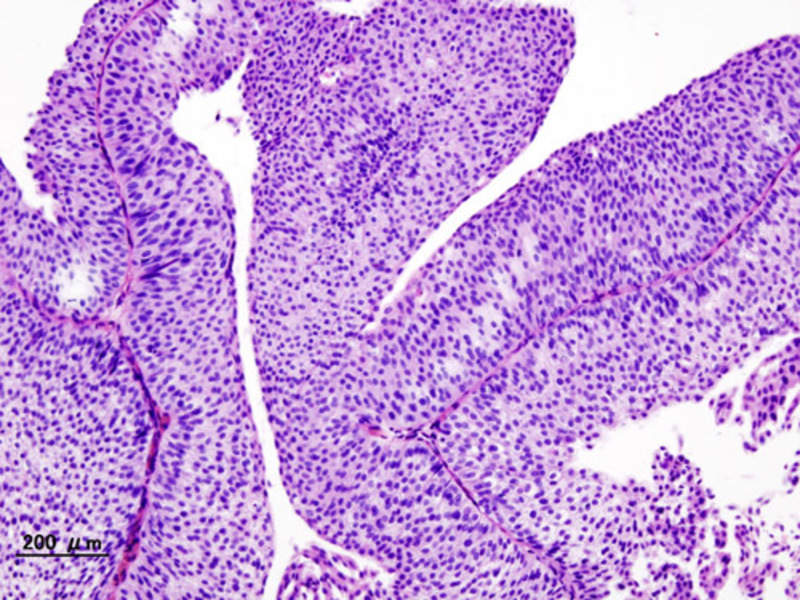
Astrazeneca S Imfinzi Receives Fda Accelerated Approval For Urothelial Fda’s latest approval of imfinzi (durvalumab) in combination with chemotherapy for treating resectable non small cell lung cancer before and after surgery is based on phase iii trial results showing that the regimen reduced the risk of recurrence, progression, or death by 32%. Durvalumab (imfinzi; astratzeneca) in combination with chemotherapy has been approved by the fda for the treatment of adult patients with resectable early (iia iiib) non small cell lung cancer (nsclc) and no known epidermal growth factor receptor (egfr) mutations or anaplastic lymphoma kinase (alk) rearrangements. The approval by the food and drug administration (fda) was based on positive results from the pivotal aegean trial, which were published in the new england journal of medicine in october 2023. Astrazeneca is backing imfinzi’s small cell lung cancer fda bid with data from the phase iii adriatic study, which showed that imfinzi could lower the risk of death by 27% in these patients as compared with placebo. the pd l1 blocker also reduced the risk of disease progression or death by 24%.

Us Fda Approves Astrazeneca S Imfinzi Combination For Biliary Tract Cancer The approval by the food and drug administration (fda) was based on positive results from the pivotal aegean trial, which were published in the new england journal of medicine in october 2023. Astrazeneca is backing imfinzi’s small cell lung cancer fda bid with data from the phase iii adriatic study, which showed that imfinzi could lower the risk of death by 27% in these patients as compared with placebo. the pd l1 blocker also reduced the risk of disease progression or death by 24%.

Astrazeneca S Imfinzi Gains Us Approval Boosting Treatment Options For
Comments are closed.