Fda Dietary Supplements Clear Compliance Guide
Dietary Supplement Regulations Guide Gmp Fda Compliance Fda issues guidance documents containing nonbinding recommendations to help industry understand and comply with all regulations. Master fda and healthcare laws for dietary supplements. ensure compliance with labeling and claims to thrive in the booming dietary market.

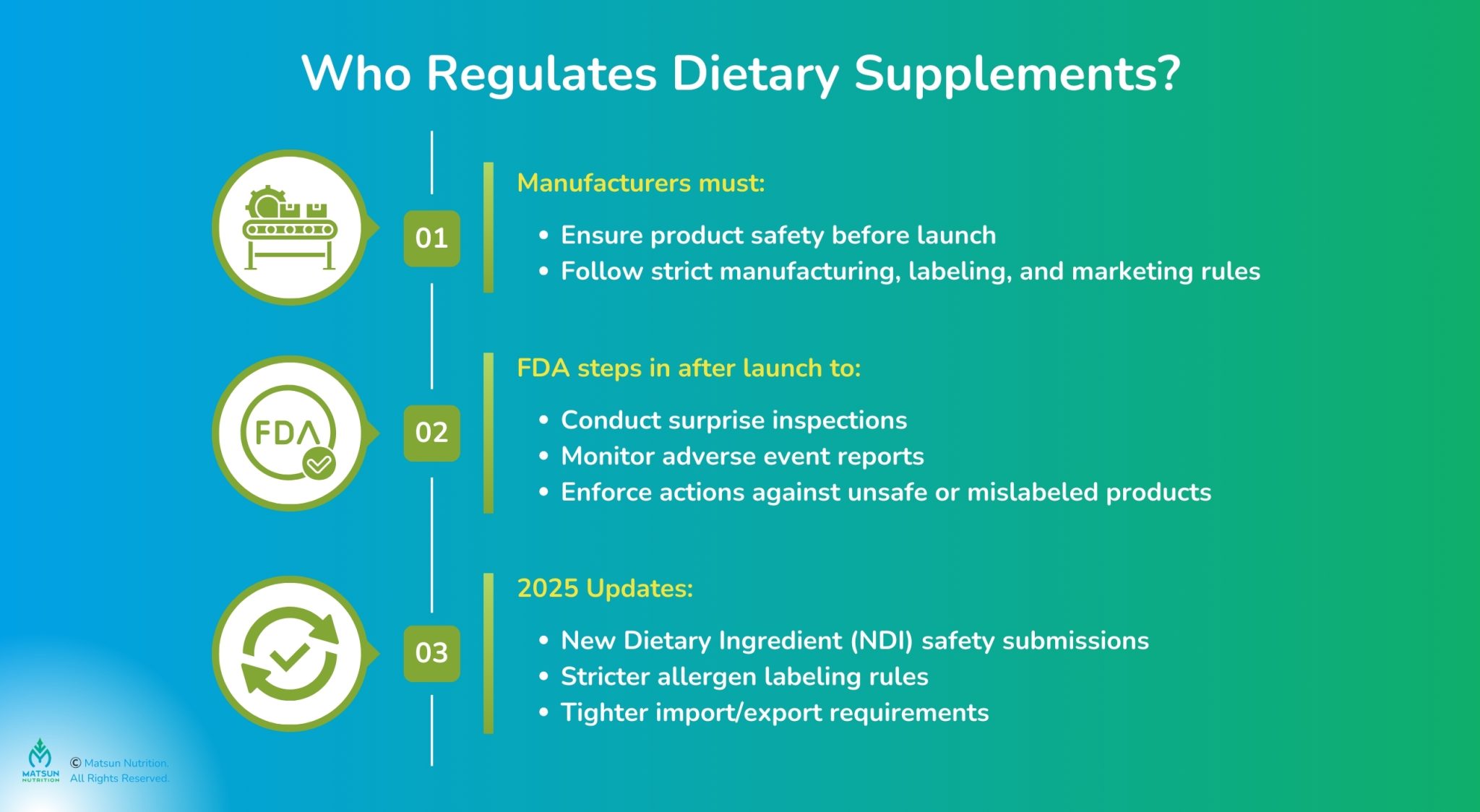
Fda Supplement Brand Guidelines Key Compliance Rules Matsun Nutrition This article provides a detailed overview of fda’s dietary supplement compliance system, highlighting key regulatory requirements, manufacturing obligations, and inspection strategies for 2026. Whether you’re launching your first formula or scaling a multi product brand, understanding how the fda regulates dietary supplements is essential to staying compliant, protecting your customers, and building long term credibility. Navigate complex fda 21 cfr part 111 regulations with ease. our guide covers mandatory labeling, ingredient safety, and market ready documentation for supplement brands. In july 2011, we published a draft guidance entitled “guidance for industry: dietary supplements: new dietary ingredient notifications and related issues,” in compliance with our statutory requirements under section 113(b) of the fda food safety modernization act (pub. l. 111 353).

Fda Supplement Brand Guidelines Key Compliance Rules Matsun Nutrition Navigate complex fda 21 cfr part 111 regulations with ease. our guide covers mandatory labeling, ingredient safety, and market ready documentation for supplement brands. In july 2011, we published a draft guidance entitled “guidance for industry: dietary supplements: new dietary ingredient notifications and related issues,” in compliance with our statutory requirements under section 113(b) of the fda food safety modernization act (pub. l. 111 353). This article will give you a clear, step by step roadmap to navigate all supplement claim rules, ensuring your label is fda compliant. if you’re unsure of how to get started on your supplements labeling, book a demo to understand how food label maker can fit your specific needs. Learn about dietary supplement labeling complete fda compliance manual in our comprehensive fda compliance guide for food labeling requirements. Understand fda rules and compliance for dietary supplements to ensure safety and legality in your products. Use the fda’s dietary supplement labeling guide and the cosm portal to plan claims and assemble files. fda doesn’t preapprove labels; instead, self check, file any claim notice within 30 days via cosm, and submit an ndi notice 75 days before marketing if an ingredient is new.
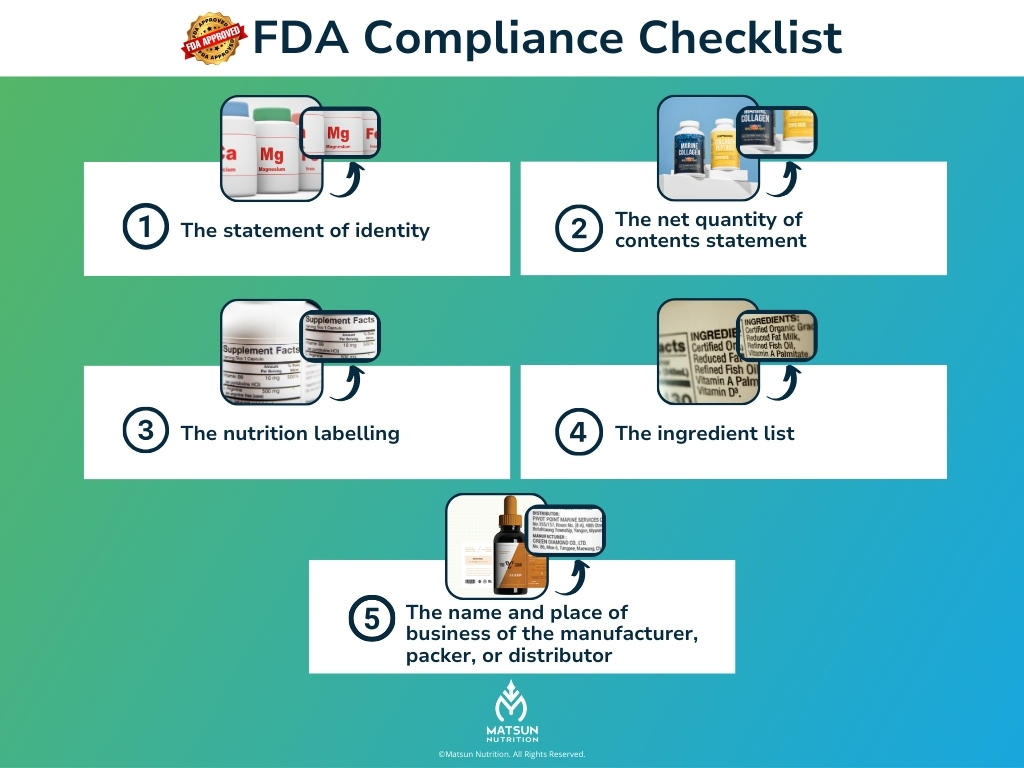
Supplement Label Requirements Fda Design Guide Matsun This article will give you a clear, step by step roadmap to navigate all supplement claim rules, ensuring your label is fda compliant. if you’re unsure of how to get started on your supplements labeling, book a demo to understand how food label maker can fit your specific needs. Learn about dietary supplement labeling complete fda compliance manual in our comprehensive fda compliance guide for food labeling requirements. Understand fda rules and compliance for dietary supplements to ensure safety and legality in your products. Use the fda’s dietary supplement labeling guide and the cosm portal to plan claims and assemble files. fda doesn’t preapprove labels; instead, self check, file any claim notice within 30 days via cosm, and submit an ndi notice 75 days before marketing if an ingredient is new.
Comments are closed.