Fda Classification System

Us Fda Medical Device Classification 1668938902 Pdf The food and drug administration (fda) has established classifications for approximately 1,700 different generic types of devices and grouped them into 16 medical specialties referred to as. Complete guide to fda device classes i, ii & iii. learn classification criteria, regulatory pathways, examples & requirements for 2025.

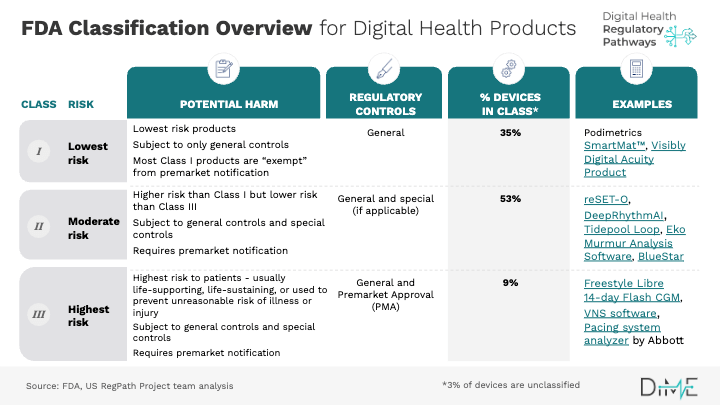
Fda Medical Device Classification Class I Ii Iii Explained 2025 Classification regulation means a section under parts 862 through 892 of this chapter that contains the identification (general description and intended use) and classification (class i, ii or iii) of a single device type or more than one related device type (s). This database contains device names and their associated product codes. the name and product code identify the generic category of a device for fda. In 1976, the medical device amendments were added to the united states (u.s.) federal food, drug and cosmetic act (fd&c act) of 1938, establishing the current risk based classification system for all medical devices the fda regulates. Medical devices are regulated by the u.s. food and drug administration (fda). each device must meet rules based on its risk to users. these rules fall into three levels: class i, class ii, and class iii. the higher the class, the more control and testing is required.
Fda Classification System In 1976, the medical device amendments were added to the united states (u.s.) federal food, drug and cosmetic act (fd&c act) of 1938, establishing the current risk based classification system for all medical devices the fda regulates. Medical devices are regulated by the u.s. food and drug administration (fda). each device must meet rules based on its risk to users. these rules fall into three levels: class i, class ii, and class iii. the higher the class, the more control and testing is required. Learn about fda's three medical device classes — class i (low risk), class ii (moderate risk), and class iii (high risk) — including examples, regulatory pathways, and how to classify your device. Learn us fda medical device classification, different device classes, the approval pathway, and how qms software facilitates the approval process. Device classification operates through a systematic evaluation of intended use, indications for use, and the level of control necessary to provide reasonable assurance of safety and effectiveness. the fda assigns each device to one of three classes based on the risk level associated with its use. When bringing a new medical device to market in the united states, one of the first—and most critical—steps is determining its classification. this step is crucial because it dictates the regulatory pathway for fda approval, as well as the level of regulatory oversight your device will require.

Fda Classification System Learn about fda's three medical device classes — class i (low risk), class ii (moderate risk), and class iii (high risk) — including examples, regulatory pathways, and how to classify your device. Learn us fda medical device classification, different device classes, the approval pathway, and how qms software facilitates the approval process. Device classification operates through a systematic evaluation of intended use, indications for use, and the level of control necessary to provide reasonable assurance of safety and effectiveness. the fda assigns each device to one of three classes based on the risk level associated with its use. When bringing a new medical device to market in the united states, one of the first—and most critical—steps is determining its classification. this step is crucial because it dictates the regulatory pathway for fda approval, as well as the level of regulatory oversight your device will require.

Fda Classification System Device classification operates through a systematic evaluation of intended use, indications for use, and the level of control necessary to provide reasonable assurance of safety and effectiveness. the fda assigns each device to one of three classes based on the risk level associated with its use. When bringing a new medical device to market in the united states, one of the first—and most critical—steps is determining its classification. this step is crucial because it dictates the regulatory pathway for fda approval, as well as the level of regulatory oversight your device will require.
Fda Classification System
Comments are closed.