Fda Approves Anaphylaxis Nasal Spray
;height:auto;width:100%25)
Fda Approves First Nasal Spray For Allergic Reactions The Washington Post Today, the u.s. food and drug administration approved neffy (epinephrine nasal spray) for the emergency treatment of allergic reactions (type i), including those that are life threatening. The food and drug administration (fda) has approved neffy epinephrine nasal spray from ars pharmaceuticals for the emergency treatment of anaphylaxis in people weighing at least 66 pounds. it is the first treatment that does not require an injection.

Fda Approves Anaphylaxis Nasal Spray Wkrn News 2 The first nasal spray for emergency treatment of type 1 allergic reactions, including anaphylaxis, in adults and in children who weigh at least 30 kilograms was approved by fda on aug. 9, 2024 (1,2). The fda approved neffy (ars pharmaceuticals), an epinephrine nasal spray for the emergency treatment of allergic reactions, including anaphylaxis, for adults and pediatric individuals who weigh at least 30 kg. On august 9, 2024, the u.s. food and drug administration (fda) approved neffy (ars pharmaceuticals, inc, san diego, calif), a nasal spray containing epinephrine for the treatment of allergic reactions in an emergency setting [1]. Many consumers are familiar with epipens and other epinephrine injectables when treating severe allergic reactions. now, the food and drug administration (fda) has approved the first non injectable.
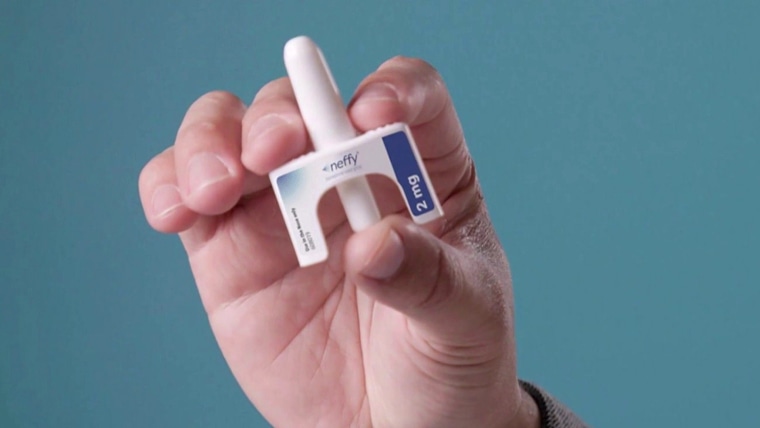
Fda Approves Nasal Spray As First Needle Free Treatment For Anaphylaxis On august 9, 2024, the u.s. food and drug administration (fda) approved neffy (ars pharmaceuticals, inc, san diego, calif), a nasal spray containing epinephrine for the treatment of allergic reactions in an emergency setting [1]. Many consumers are familiar with epipens and other epinephrine injectables when treating severe allergic reactions. now, the food and drug administration (fda) has approved the first non injectable. The u.s. food and drug administration approved neffy®, the first ever nasal spray form of epinephrine, offering a needle free alternative for treating severe, life threatening allergic reactions, or anaphylaxis. Jama. 2024 oct 1;332 (13):1043. doi: 10.1001 jama.2024.17605. "today's approval provides the first epinephrine product for the treatment of anaphylaxis that is not administered by injection. The fda approved neffy 1 mg, an epinephrine nasal spray, for the treatment of type i allergic reactions, including anaphylaxis, for children 4 years and older who weigh 15 to less than 30 kg.
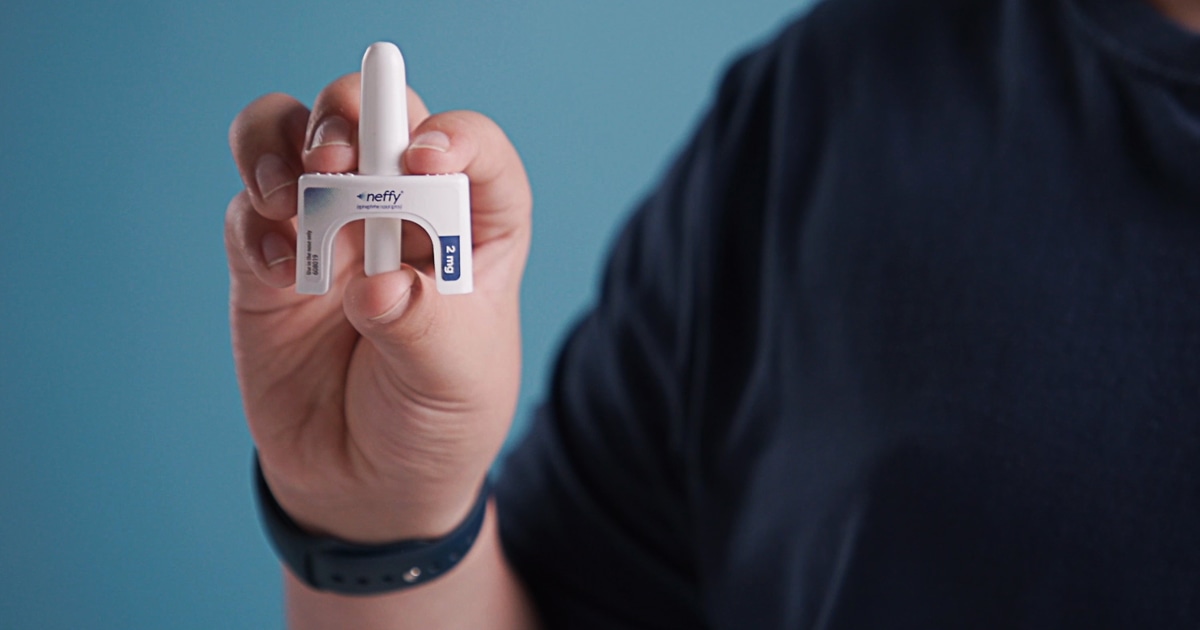
Fda Approves Nasal Spray As First Needle Free Treatment For Anaphylaxis The u.s. food and drug administration approved neffy®, the first ever nasal spray form of epinephrine, offering a needle free alternative for treating severe, life threatening allergic reactions, or anaphylaxis. Jama. 2024 oct 1;332 (13):1043. doi: 10.1001 jama.2024.17605. "today's approval provides the first epinephrine product for the treatment of anaphylaxis that is not administered by injection. The fda approved neffy 1 mg, an epinephrine nasal spray, for the treatment of type i allergic reactions, including anaphylaxis, for children 4 years and older who weigh 15 to less than 30 kg.
Neffy S New Treatment For Anaphylaxis Gets Fda Approval Fast Company "today's approval provides the first epinephrine product for the treatment of anaphylaxis that is not administered by injection. The fda approved neffy 1 mg, an epinephrine nasal spray, for the treatment of type i allergic reactions, including anaphylaxis, for children 4 years and older who weigh 15 to less than 30 kg.
Comments are closed.