Fda Approval Pathways 101

Fda Approval Pathways Devices Get to know fda’s drug development and approval process ensuring that drugs work and that the benefits outweigh their known risks. Requires fda to specify conditions for required post approval studies for drugs approved under accelerated approval by the time the drug is approved. may include enrollment targets and milestones, including the target date for study completion.

Fda Approval Pathways Devices The process of gaining fda approval to market a new medicinal product, beginning with the submission of a “new drug application” has since undergone a series of updates and amendments but, even today, remains consistent with the 1938 regulations as originally outlined. This article provides a step by step guide to the fda submission and approval process, integrating statutory references, procedural insights, and strategic considerations for global compliance. In this post, we will understand the fda approval process step by step from preclinical testing to post market surveillance and help you navigate this complex regulatory process with clarity. Learn how the fda drug approval process works, including key steps, timelines, clinical trials, and what to expect before a drug reaches patients.

Fda Approval Pathways Devices In this post, we will understand the fda approval process step by step from preclinical testing to post market surveillance and help you navigate this complex regulatory process with clarity. Learn how the fda drug approval process works, including key steps, timelines, clinical trials, and what to expect before a drug reaches patients. This guide explains how the fda approval process works from start to finish — the submissions, review types, decision points, and pathways that shape drug development strategy and create the catalysts that move markets. In this comprehensive post, we will walk you through the fda approval journey step by step, from pre clinical testing to post market surveillance, helping you navigate this critical regulatory landscape with clarity. In this paper, we discuss the various opportunities the us food and drug administration and the european medicines agency offer to expedite the drug development and regulatory approval timelines for drugs intended to treat serious diseases and unmet medical needs. In this guide, we’ll walk you through each step of the fda drug approval process, from discovery to patient care, providing key insights into clinical trials, regulatory submissions, and fda reviews.
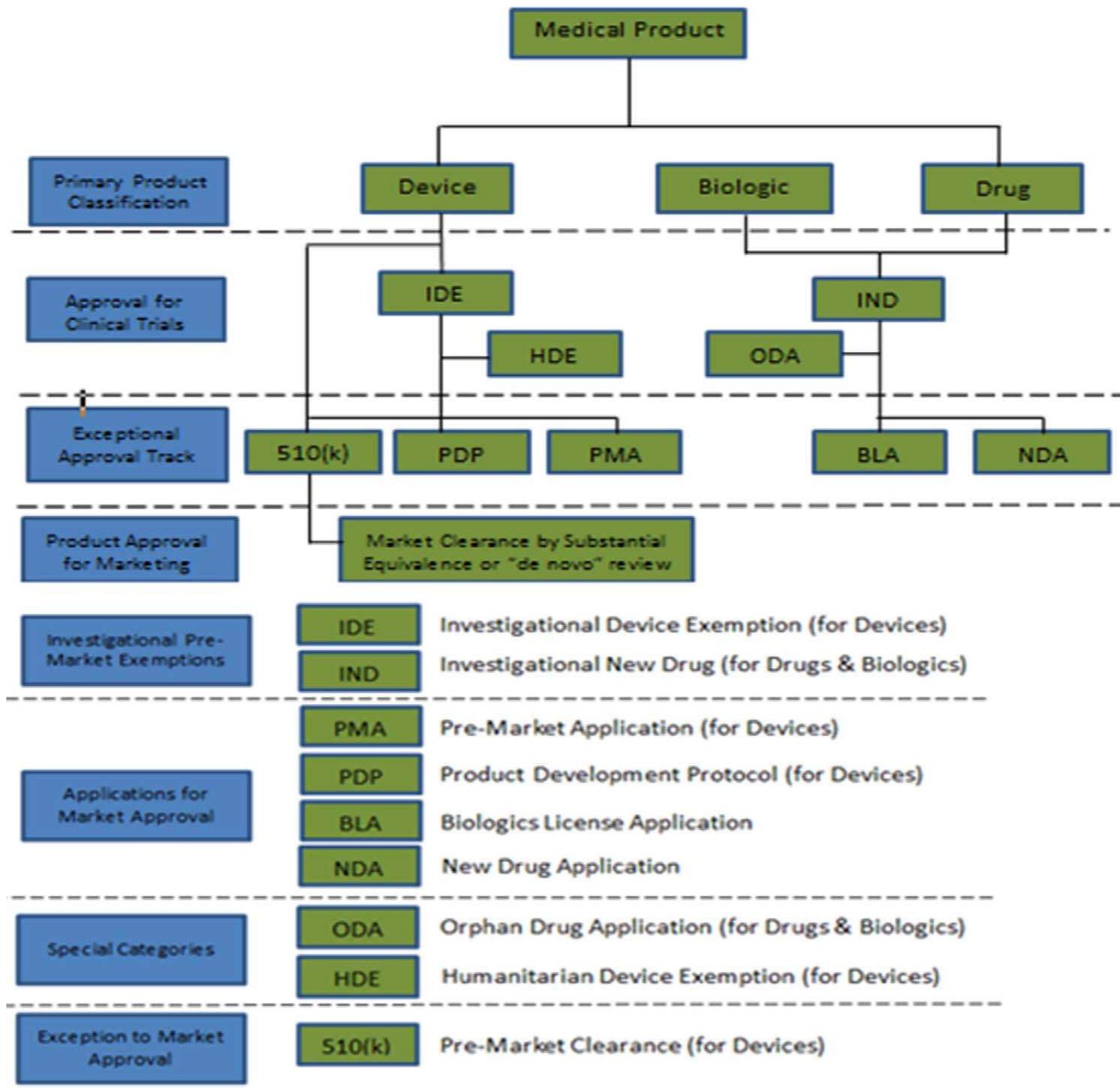
Review And Approval Pathways By The U S Fda Product This guide explains how the fda approval process works from start to finish — the submissions, review types, decision points, and pathways that shape drug development strategy and create the catalysts that move markets. In this comprehensive post, we will walk you through the fda approval journey step by step, from pre clinical testing to post market surveillance, helping you navigate this critical regulatory landscape with clarity. In this paper, we discuss the various opportunities the us food and drug administration and the european medicines agency offer to expedite the drug development and regulatory approval timelines for drugs intended to treat serious diseases and unmet medical needs. In this guide, we’ll walk you through each step of the fda drug approval process, from discovery to patient care, providing key insights into clinical trials, regulatory submissions, and fda reviews.
Comments are closed.