Fda And Medical Device Reporting

Adverse Event Reporting For Medical Devices Complete Fda Medical Medical device reporting (mdr) is one of the postmarket surveillance tools the fda uses to monitor device performance, detect potential device related safety issues, and contribute to. (1) if you are a health professional or consumer or other entity, you may submit voluntary reports to fda regarding devices or other fda regulated products using the form fda 3500.
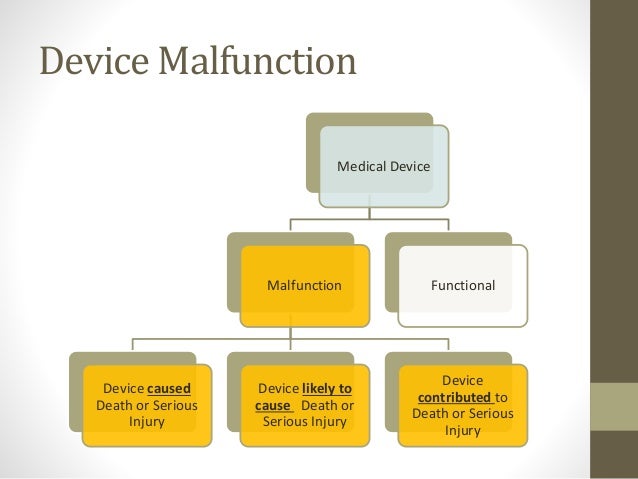
Fda And Medical Device Reporting The federal food, drug, and cosmetic act, section 519 (records and reports on devices) grants the fda authority to require mandatory medical device reports from:. Medical device reporting (mdr) under 21 cfr part 803 is how the u.s. food and drug administration (fda) collects reports when a medical device is linked to a death, a serious injury, or a malfunction that could cause harm if it happens again. This guidance document describes and explains the food and drug administration’s (fda, we, us) current regulation that addresses reporting and recordkeeping requirements applicable to. Medical device reporting (mdr) regulations require manufacturers to report to fda device related incidents, deaths, serious injuries, and device malfunctions which are likely to cause or contribute to death or serious injury if they were to occur.

4 Fda Medical Device Reporting Procedures For Life Science Companies This guidance document describes and explains the food and drug administration’s (fda, we, us) current regulation that addresses reporting and recordkeeping requirements applicable to. Medical device reporting (mdr) regulations require manufacturers to report to fda device related incidents, deaths, serious injuries, and device malfunctions which are likely to cause or contribute to death or serious injury if they were to occur. Patients, healthcare professionals and consumers who find a problem related to a medical device are encouraged to report medical device adverse events or product problems to the fda through medwatch, the fda safety information and adverse event reporting program. A new government accountability office (gao) report has uncovered concerning issues with the fda's handling of dangerous medical device recalls. despite its authority, the fda rarely takes decisive action to remove defective devices from the market, leaving patients vulnerable to potential harm. the report attributes this to a severe lack of resources and staff at the agency, raising questions. This article will review the medical device reporting (mdr) requirements, important stakeholders, the process required for reporting a medical device problem as a medical device manufacturer or user, and the critical deadlines for medical device reports. Learning objectives describe fda’s regulatory authority for medical device reporting define medical device reporting terms identify mandatory reporters and their responsibilities determine how, when, and where to report.

Fda Medical Device Reporting Patients, healthcare professionals and consumers who find a problem related to a medical device are encouraged to report medical device adverse events or product problems to the fda through medwatch, the fda safety information and adverse event reporting program. A new government accountability office (gao) report has uncovered concerning issues with the fda's handling of dangerous medical device recalls. despite its authority, the fda rarely takes decisive action to remove defective devices from the market, leaving patients vulnerable to potential harm. the report attributes this to a severe lack of resources and staff at the agency, raising questions. This article will review the medical device reporting (mdr) requirements, important stakeholders, the process required for reporting a medical device problem as a medical device manufacturer or user, and the critical deadlines for medical device reports. Learning objectives describe fda’s regulatory authority for medical device reporting define medical device reporting terms identify mandatory reporters and their responsibilities determine how, when, and where to report.

Preparing For Fda Inspections Of Medical Device Reporting Mdr This article will review the medical device reporting (mdr) requirements, important stakeholders, the process required for reporting a medical device problem as a medical device manufacturer or user, and the critical deadlines for medical device reports. Learning objectives describe fda’s regulatory authority for medical device reporting define medical device reporting terms identify mandatory reporters and their responsibilities determine how, when, and where to report.
Comments are closed.