F Block Elements Inner Transition Elements Lanthanides And
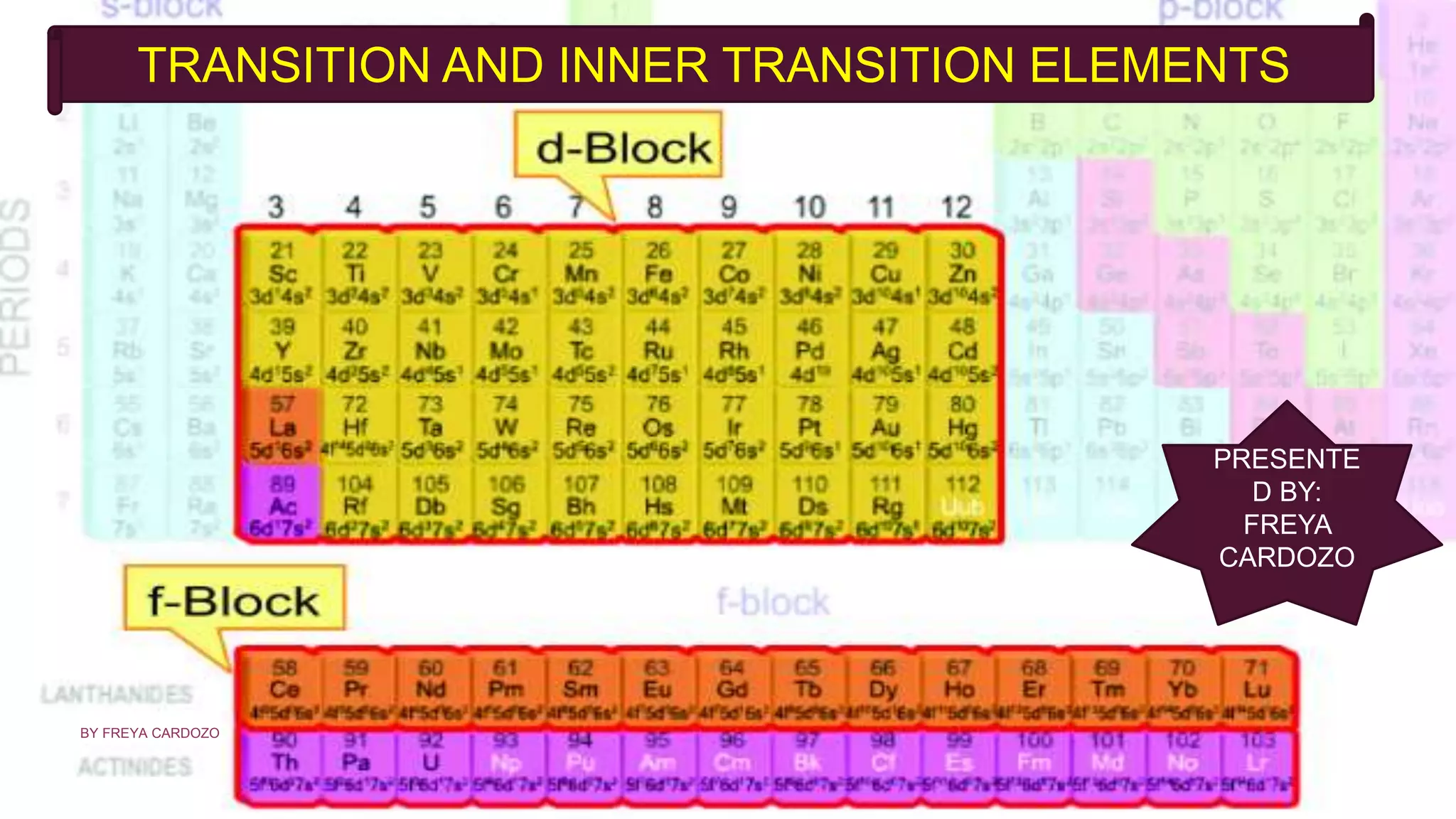
D And F Block Elements Transition And Inner Transition Elements Pptx The elements that have incompletely filled (n 2) f orbitals in their ground state or in any of their oxidation states are called f block elements or inner transition. Inner transition elements are special elements found in the f block of the periodic table. they include two series: the lanthanides (atomic numbers 58–71) and actinides (atomic numbers 90–103).
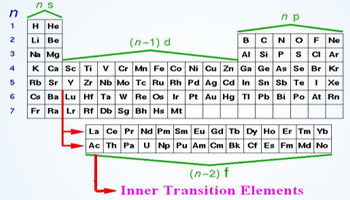
Where Are The Inner Transition Elements Infoupdate Org These elements are formal members of group 3 but are shown separately as f block of the periodic table. the elements of f block are also called inner transition elements. the two series of inner transition elements that are 4f and 5f series are known as lanthanoids and actinoids respectively. F block elements, also known as inner transition elements encompassing both lanthanides and actinides, exhibit a wide range of applications across different scientific, industrial, and technological domains. Learn about the f block elements in chemistry, which are also the inner transition metals. discover their properties and uses. The f block elements, also known as inner transition metals, are found in the two series located at the bottom of the periodic table. these elements belong to the lanthanide and actinide series, distinguished by their placement within the f orbital block.

Which Block Has Inner Transition Elements The F Block Guide Learn about the f block elements in chemistry, which are also the inner transition metals. discover their properties and uses. The f block elements, also known as inner transition metals, are found in the two series located at the bottom of the periodic table. these elements belong to the lanthanide and actinide series, distinguished by their placement within the f orbital block. F block elements, also called inner transition elements, are those in which the (n–2)f orbitals (inner penultimate shell) are progressively filled. they are placed separately at the bottom of the periodic table and consist of two series — lanthanides (4f) and actinides (5f). The f block elements are found in two rows placed separately at the bottom of the periodic table, known as the lanthanides and actinides. This document discusses the inner transition elements known as f block or f elements, which include the lanthanides and actinides. it provides historical context on their discovery, noting many were initially thought to be single elements but were later found to be mixtures. Due to lanthanide contraction, the elements of 5d (third) and 4d (second) transition series resemble each other much more closely than do the elements of 4d (second) and 3d (first) series.

D And F Block Elements Transition And Inner Transition Elements Pptx F block elements, also called inner transition elements, are those in which the (n–2)f orbitals (inner penultimate shell) are progressively filled. they are placed separately at the bottom of the periodic table and consist of two series — lanthanides (4f) and actinides (5f). The f block elements are found in two rows placed separately at the bottom of the periodic table, known as the lanthanides and actinides. This document discusses the inner transition elements known as f block or f elements, which include the lanthanides and actinides. it provides historical context on their discovery, noting many were initially thought to be single elements but were later found to be mixtures. Due to lanthanide contraction, the elements of 5d (third) and 4d (second) transition series resemble each other much more closely than do the elements of 4d (second) and 3d (first) series.
Comments are closed.