Evolving Functional Intrinsically Disordered Proteins Through Directed
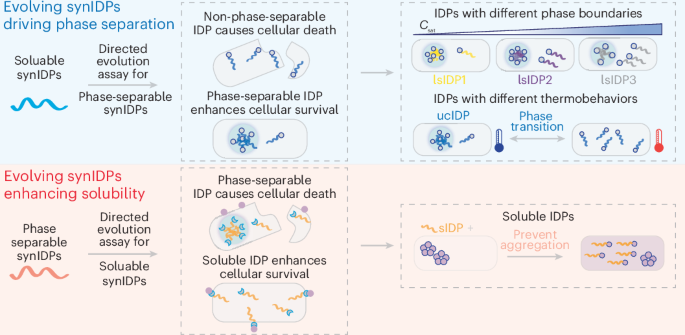
Directed Evolution Of Functional Intrinsically Disordered Proteins Here, to circumvent this design challenge, we present a systematic directed evolution method for the evolution of synidps capable of mediating diverse phase behaviors in living cells. Here, to circumvent this design challenge, we present a systematic directed evolution method for the evolution of synidps capable of mediating diverse phase behaviors in living cells.

Video Intrinsically Disordered Proteins Here, to circumvent this design challenge, we present a systematic directed evolution method for the evolution of synidps capable of mediating diverse phase behaviors in living cells. Subject of research: directed evolution of synthetic intrinsically disordered proteins (synidps) for phase behavior regulation and antibiotic resistance reversal. This review focuses on different aspects of disordered proteins and disordered protein regions, which form the basis of the so called “disorder–function paradigm” of proteins. Here we review these recent advances with a focus on the dynamics and interactions of idps. it has been 25 years since the concept of intrinsically disordered proteins (idps) was proposed [1].

Research Frontiers Of Intrinsically Disordered Proteins This review focuses on different aspects of disordered proteins and disordered protein regions, which form the basis of the so called “disorder–function paradigm” of proteins. Here we review these recent advances with a focus on the dynamics and interactions of idps. it has been 25 years since the concept of intrinsically disordered proteins (idps) was proposed [1]. Yifan dai and his team designed a method based on directed evolution to create synthetic intrinsically disordered proteins that can facilitate diverse phase behaviors in living cells. Design of functional intrinsically disordered proteins relies on a combination of computational tools and heuristics gleaned from sequence function studies. there are few cases where intrinsically disordered proteins have made it into industrial applications. Here, we use multinuclear chemical exchange saturation (cest) nuclear magnetic resonance to determine the structure of a thermally accessible globally folded excited state in equilibrium with the intrinsically disordered native ensemble of a bacterial transcriptional regulator cytr.
Comments are closed.