Evaluating The Interchangeability Of Hrd Testing In Ovarian Cancer
Homologous Recombination Deficiency In Ovarian Cancer From The This review summarizes the genomic and functional hrd assays currently used in clinical practice and those under evaluation, the clinical implications of hrd testing in oc, and their current pitfalls and limitations. Assessment of homologous recombination deficiency (hrd) status is now essential for ovarian cancer patient management. the aim of our study was to analyze the influence of ethnic variations, tumor purity, and neoadjuvant chemotherapy (ct) on the.
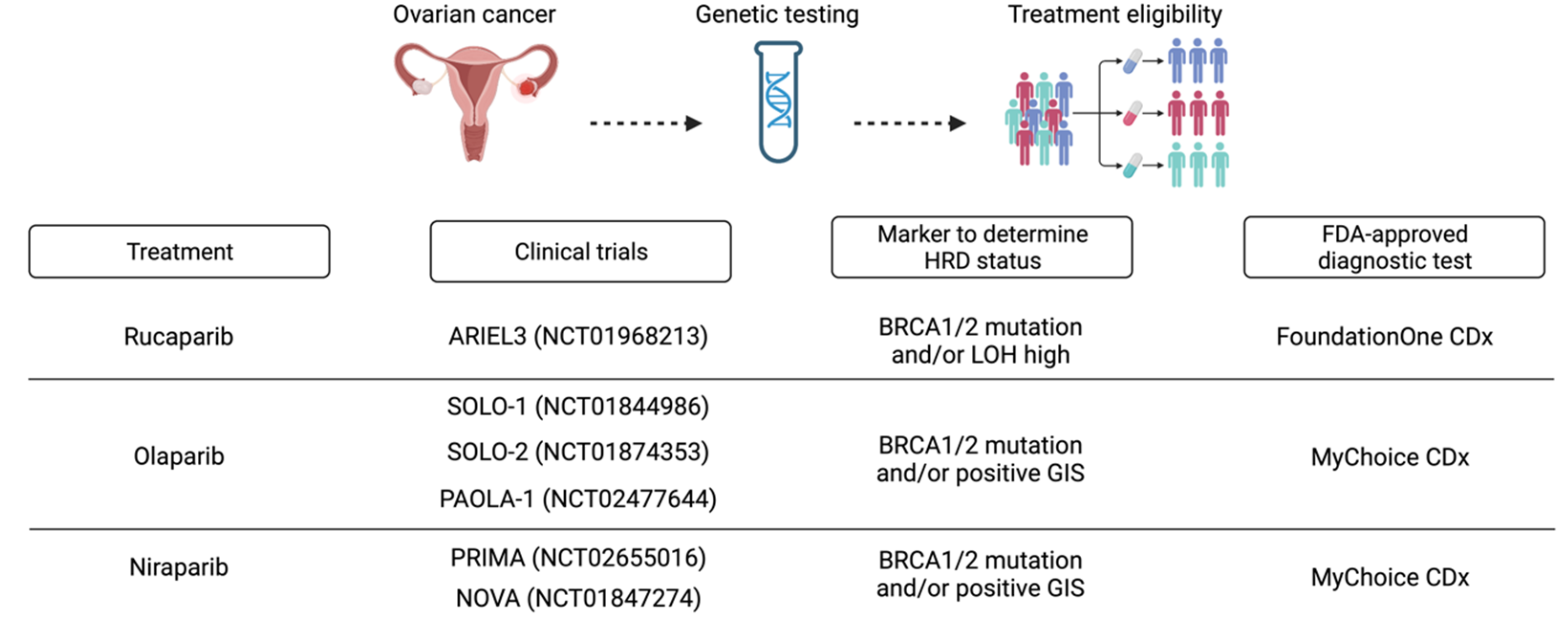
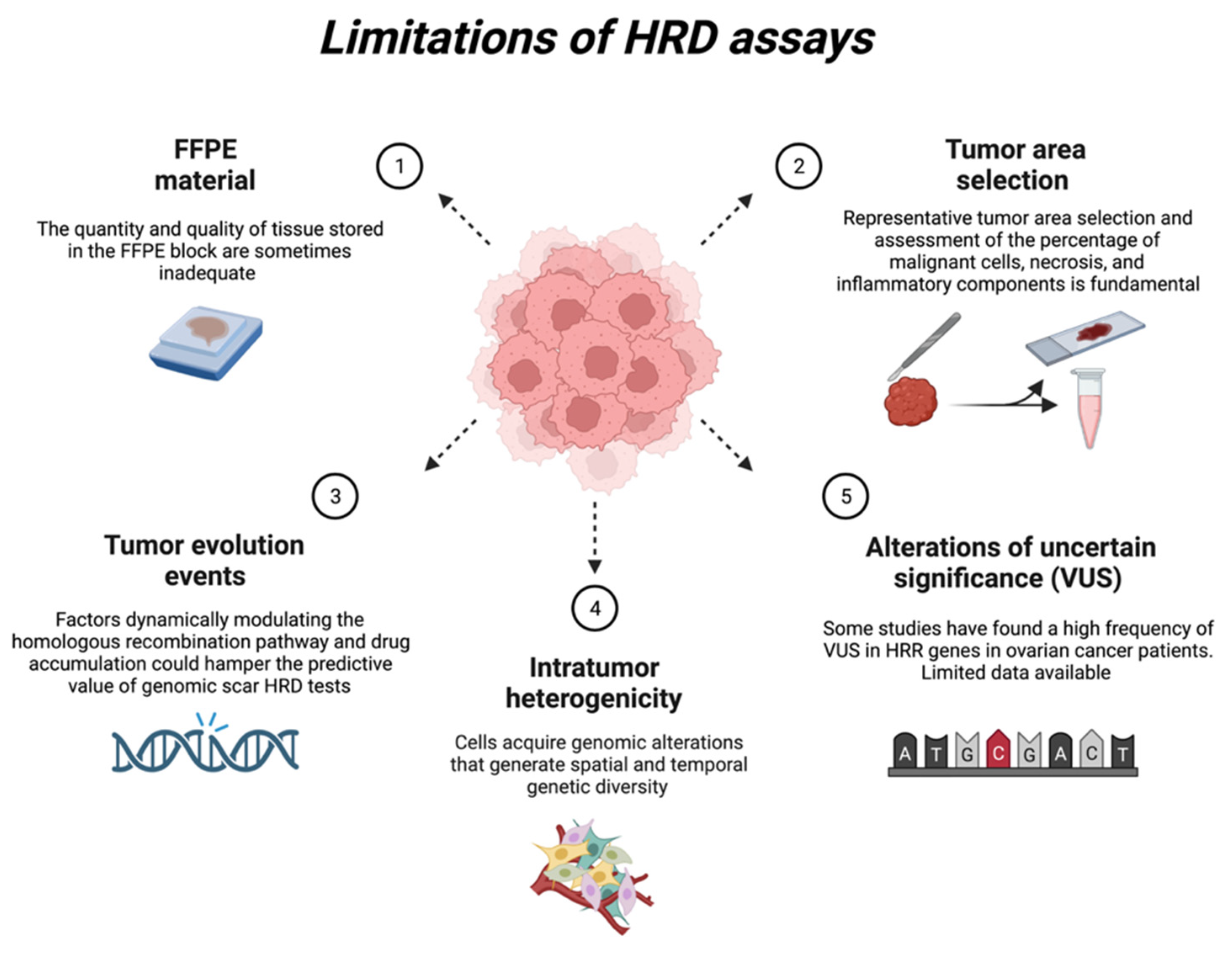
Homologous Recombination Deficiency In Ovarian Cancer From The Benoit you, md, phd, lyon university hospital, lyon, france, comments on whether the various tests for homologous recombination deficiency (hrd) are comparable in ovarian cancer. hrd testing is necessary to optimize treatment, but each test will have different validation assays. We aimed to extensively compare available hrd cdx and establish a worldwide expert consensus on hrd testing in primary and recurrent oc. a group of 99 global experts from 31 different countries was formed. However, hrd assays also have several limitations in identifying hrd positive ovarian cancer, including issues with the mychoice® gis cutoff value, the existence of alternative evaluation methods other than mychoice®, and problems related to tumor heterogeneity and changing hrd score over time. Recent advances in high grade serous ovarian carcinoma (hgsc) treatment have been driven by the availability of clinical testing for homologous recombination deficiency (hrd), determining patients who most benefit from maintenance therapy with poly (adp ribose) polymerase inhibitors (parpi).

Full Article Homologous Recombination Deficiency Testing In Patients However, hrd assays also have several limitations in identifying hrd positive ovarian cancer, including issues with the mychoice® gis cutoff value, the existence of alternative evaluation methods other than mychoice®, and problems related to tumor heterogeneity and changing hrd score over time. Recent advances in high grade serous ovarian carcinoma (hgsc) treatment have been driven by the availability of clinical testing for homologous recombination deficiency (hrd), determining patients who most benefit from maintenance therapy with poly (adp ribose) polymerase inhibitors (parpi). This review briefly outlines the biological basis of hrd and its clinical significance and then focuses on currently available assays for hrd assessment. Here, we compare the performance of a multitude of alternative assays in comparison with myriad mychoice in high grade serous ovarian cancer (hgsoc). Ovarian cancer (ovc) constitutes significant management challenges primarily due to its late stage diagnosis and the development of resistance to chemotherapy. Therefore, this enygo survey aims to evaluate current practices and barriers in hrd and brca testing to improve equitable access to testing and personalized therapies in ovarian cancer.
Comments are closed.