Eu Mdr Device Classification Guide Advisera

Eu Mdr Medical Device Classifications Pdf Explore how to classify medical devices under mdr, including class 1, 2, and 3 devices. Viii of regulation (eu) 2017 745 on medical devices (mdr). they correspond, to a large extent, to the classification rules established by the international medical device regulators.
Eu Mdr Device Classification We're mdlaw, the leading database on european mdr and ivdr legal and regulatory information. we'll elevate your medical devices into full eu compliance. Casus consulting’s classification tool is intended to provide users with an understanding of the likely classification for a specific medical device, as a starting point to entering the eu market. If you're looking for mdr examples, check out our list of mdr example devices and their relevant eu mdr classification. Uncertain about what products are subject to medical device classification? learn how to classify medical devices under the eu mdr here.

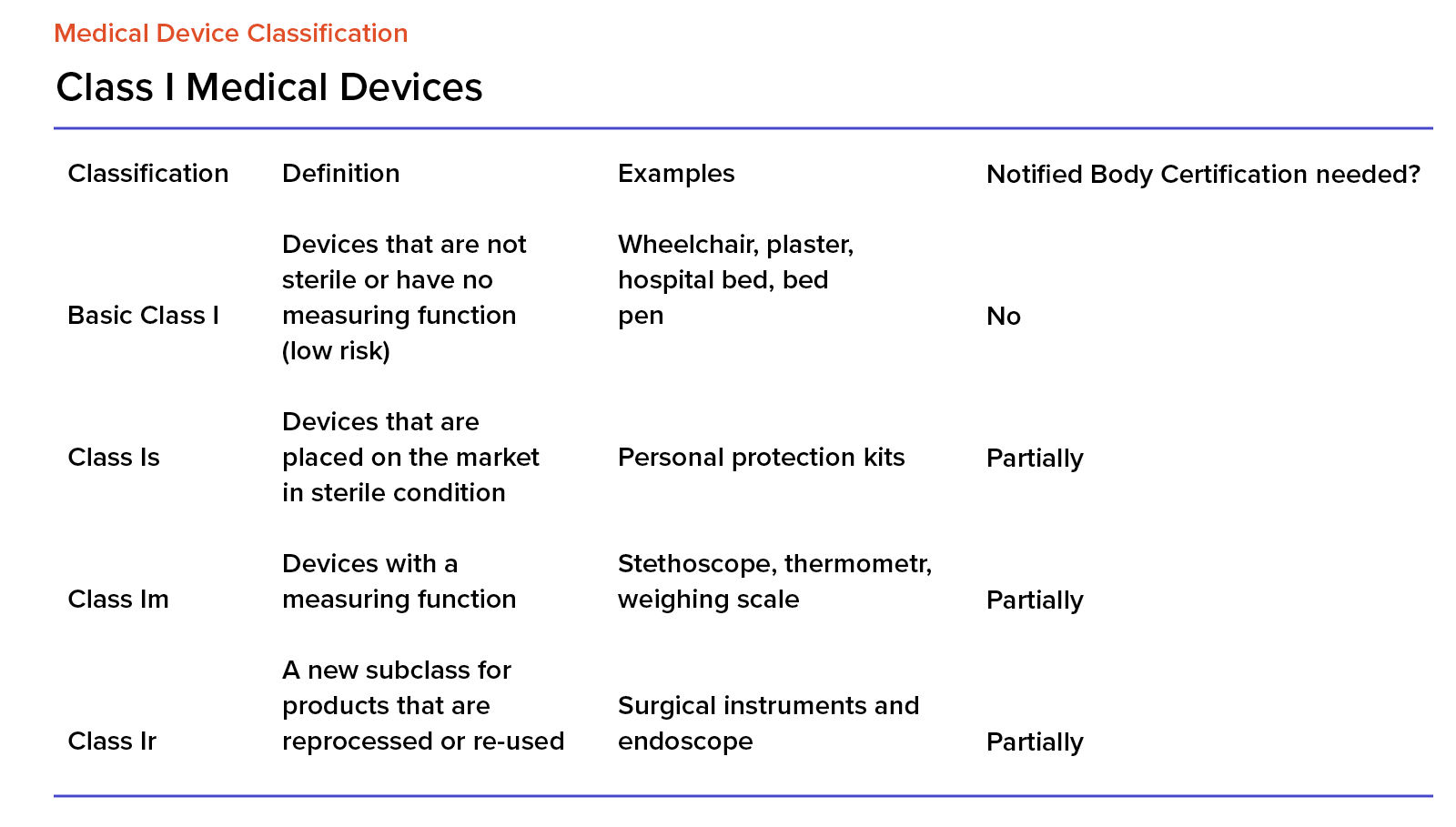
Medical Device Classification In Europe Your Guide To Navigating If you're looking for mdr examples, check out our list of mdr example devices and their relevant eu mdr classification. Uncertain about what products are subject to medical device classification? learn how to classify medical devices under the eu mdr here. A complete guide to the european commission's december 2025 proposal to simplify mdr and ivdr. covers the new rule 11 for software, indefinite certificate validity, regulatory sandboxes, breakthrough and orphan device pathways, fixed conformity timelines, sme fee reductions, and the expected legislative timeline through 2027. Learn how eu mdr medical device classification impacts compliance across class i, ii, iii devices. streamline certification. Planning for implementation of the european union medical devices regulations – are you prepared? medical device clinical investigations – what’s new under the mdr? how to get to the market and stay there! do you know the requirements and your responsibilities for medical device vigilance reporting?. In this guide, we present the different classes for medical devices, explain how medical devices must be classified, delve into the classification rules, and explore the implications of each classification.
Eu Mdr Medical Device Classification Classes And Examples A complete guide to the european commission's december 2025 proposal to simplify mdr and ivdr. covers the new rule 11 for software, indefinite certificate validity, regulatory sandboxes, breakthrough and orphan device pathways, fixed conformity timelines, sme fee reductions, and the expected legislative timeline through 2027. Learn how eu mdr medical device classification impacts compliance across class i, ii, iii devices. streamline certification. Planning for implementation of the european union medical devices regulations – are you prepared? medical device clinical investigations – what’s new under the mdr? how to get to the market and stay there! do you know the requirements and your responsibilities for medical device vigilance reporting?. In this guide, we present the different classes for medical devices, explain how medical devices must be classified, delve into the classification rules, and explore the implications of each classification.
Eu Mdr Medical Device Classification Classes And Examples Planning for implementation of the european union medical devices regulations – are you prepared? medical device clinical investigations – what’s new under the mdr? how to get to the market and stay there! do you know the requirements and your responsibilities for medical device vigilance reporting?. In this guide, we present the different classes for medical devices, explain how medical devices must be classified, delve into the classification rules, and explore the implications of each classification.
Eu Mdr Classification Rules Guide To Classification Of A Medical
Comments are closed.