Ethical Challenges In Study Design And Informed Consent Ethical

Ethical Issues In Genetic Research Disclosure And Informed Consent Pdf Ten case vignettes illustrate ethical challenges that arise in international research with culturally diverse populations. recommendations for researchers and policy makers concerned about ethical practices in multinational studies conducted in resource poor settings are also listed. This chapter aims to provide an overview of significant historical milestones in the development of informed consent, current consensus on its structural features, and examples of situations.

Understanding Informed Consent An Ethical Imperative In This review considers ethical challenges to research design and informed consent in biomedical and behavioural studies conducted in resource poor settings and explores relevant social, cultural, and ethical issues in the conduct of biomedical and social health research in developing countries. Despite legislative efforts like the fdama and pediatric research equity act, ethical challenges persist in obtaining ic in pediatric studies. pediatric research necessitates nuanced ic approaches, involving parents, guardians, and children. Despite being widely acknowledged as essential, the practical implementation of informed consent is fraught with ethical complexities that challenge researchers, regulators, ethicists worldwide. Though respect for patient autonomy is central to informed consent, the process is also shaped by the ethical principles of beneficence, nonmaleficence, and justice.

Pdf Lack Of Informed Consent And Absence Of Ethical Committee Despite being widely acknowledged as essential, the practical implementation of informed consent is fraught with ethical complexities that challenge researchers, regulators, ethicists worldwide. Though respect for patient autonomy is central to informed consent, the process is also shaped by the ethical principles of beneficence, nonmaleficence, and justice. This study explored the ethical challenges encountered by research team members during the informed consent process when enrolling participants with schizophrenia and bipolar affective disorder into research. This article explores the complexities of informed consent in clinical research, highlighting key challenges and proposing potential solutions [1]. informed consent is grounded in ethical principles such as autonomy, beneficence, and justice. Thus, given the nature of dcts that may involve an informed consent process in a remote manner and or electronic methods for the signature of the informed consent form, several ethical aspects must be assessed including the trials design, its population, the risks, burdens, and potential benefits. In this piece, we focus on these ethical challenges and integrate them with practical considerations outlined above. we articulate paths forward for major types of acute cardiac care research, emphasising throughout the importance of attention to critical contextual factors (figure 1).
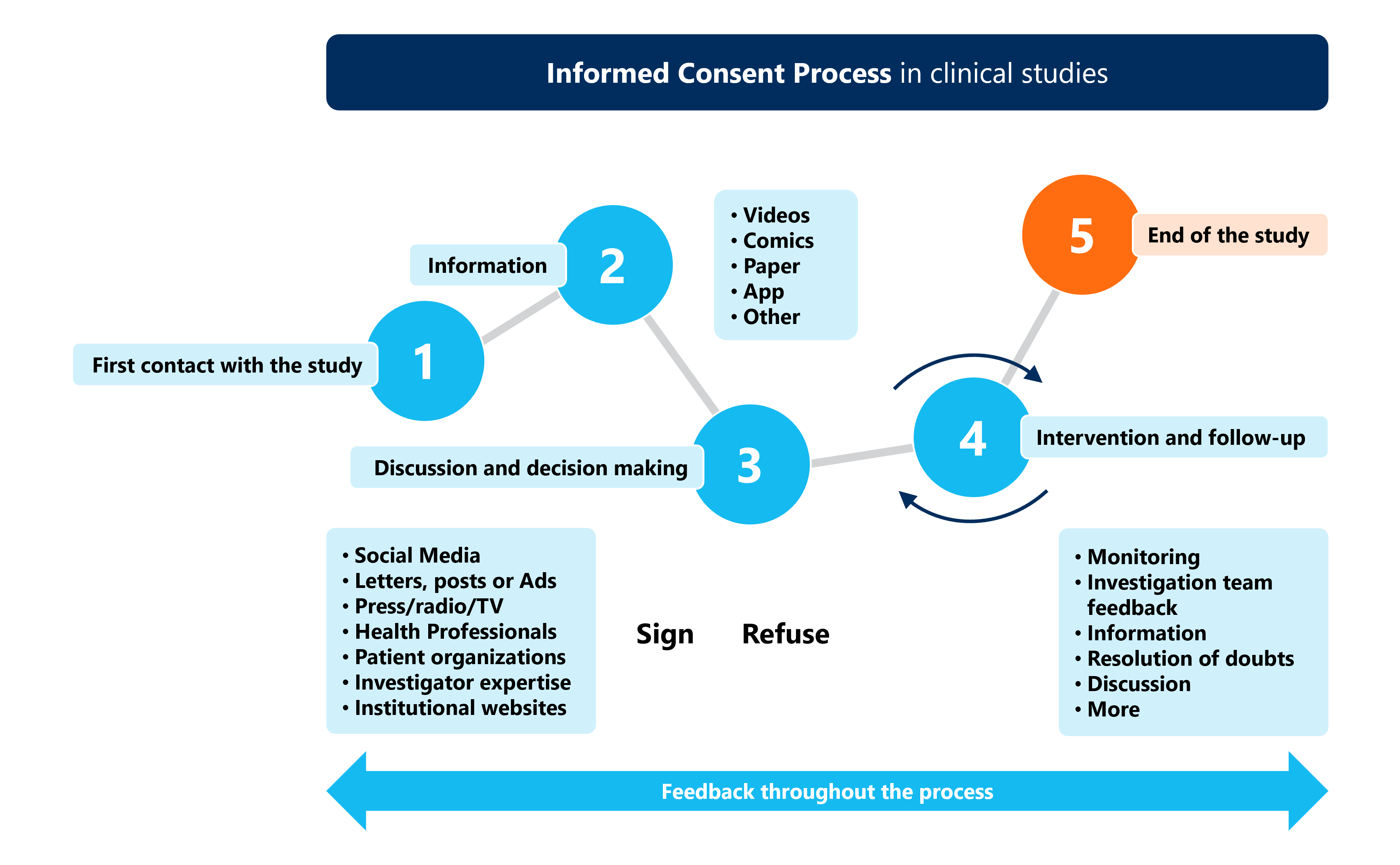
Informed Consent Comic This study explored the ethical challenges encountered by research team members during the informed consent process when enrolling participants with schizophrenia and bipolar affective disorder into research. This article explores the complexities of informed consent in clinical research, highlighting key challenges and proposing potential solutions [1]. informed consent is grounded in ethical principles such as autonomy, beneficence, and justice. Thus, given the nature of dcts that may involve an informed consent process in a remote manner and or electronic methods for the signature of the informed consent form, several ethical aspects must be assessed including the trials design, its population, the risks, burdens, and potential benefits. In this piece, we focus on these ethical challenges and integrate them with practical considerations outlined above. we articulate paths forward for major types of acute cardiac care research, emphasising throughout the importance of attention to critical contextual factors (figure 1).
Comments are closed.