Equilibrium Introduction

Equilibrium Introduction Pdf When a chemical reaction is at equilibrium, any disturbance of the system, such as a change in temperature, or addition or removal of one of the reaction components, will "shift" the composition to a new equilibrium state. This chapter defines equilibrium in mixtures and ideal solutions and introduces the equilibrium constant, its relationship with the gibbs free energy and its dependence on temperature.

Introduction To Equilibrium Theory A Quick Clickview The concept of equilibrium and the definition of an equilibrium state is of great importance in physics and chemistry. in order to gain an understanding of the principles that govern all equilibria, we'll take the approach of examining in detail a series of examples of different types of equilibria. Chemical equilibrium is the condition in the course of a reversible chemical reaction in which no net change in the amounts of reactants and products occurs. a reversible chemical reaction is one in which the products, as soon as they are formed, react to produce the original reactants. In chemistry, chemical equilibrium describes a state during a reversible reaction when the concentrations of reactants and products remain constant over time. this balance happens when the forward and reverse reactions occur at the same rate. In a chemical equilibrium, the forward and reverse reactions occur at equal rates, and the concentrations of products and reactants remain constant. a catalyst speeds up the rate of a chemical reaction, but has no effect upon the equilibrium position for that reaction.

Equilibrium Introduction Class Eleven Chemistry In chemistry, chemical equilibrium describes a state during a reversible reaction when the concentrations of reactants and products remain constant over time. this balance happens when the forward and reverse reactions occur at the same rate. In a chemical equilibrium, the forward and reverse reactions occur at equal rates, and the concentrations of products and reactants remain constant. a catalyst speeds up the rate of a chemical reaction, but has no effect upon the equilibrium position for that reaction. Stable equilibrium: a system returns to its equilibrium state after a small perturbation. unstable equilibrium: a system moves away from its equilibrium state after a small perturbation. Equilibrium is defined as the state of a system in which the measurable properties of a system like colour, concentration, pressure etc become constant. there are two types of equilibrium: (a) physical equilibrium (b) chemical equilibrium. Chemical equilibrium occurs when a reversible reaction reaches a state where the rates of the forward and reverse reactions are equal, and the concentrations of reactants and products remain constant over time. Every reaction has a point in which equilibrium is established. for many reactions, it occurs at the point when essentially all reactants are converted to products; for practical purposes, scientists say that the reaction has gone to completion.

Introduction To Equilibrium Stable equilibrium: a system returns to its equilibrium state after a small perturbation. unstable equilibrium: a system moves away from its equilibrium state after a small perturbation. Equilibrium is defined as the state of a system in which the measurable properties of a system like colour, concentration, pressure etc become constant. there are two types of equilibrium: (a) physical equilibrium (b) chemical equilibrium. Chemical equilibrium occurs when a reversible reaction reaches a state where the rates of the forward and reverse reactions are equal, and the concentrations of reactants and products remain constant over time. Every reaction has a point in which equilibrium is established. for many reactions, it occurs at the point when essentially all reactants are converted to products; for practical purposes, scientists say that the reaction has gone to completion.
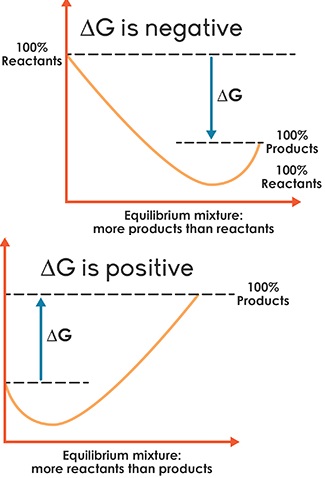
Introduction Of Equilibrium Chemical equilibrium occurs when a reversible reaction reaches a state where the rates of the forward and reverse reactions are equal, and the concentrations of reactants and products remain constant over time. Every reaction has a point in which equilibrium is established. for many reactions, it occurs at the point when essentially all reactants are converted to products; for practical purposes, scientists say that the reaction has gone to completion.
Comments are closed.