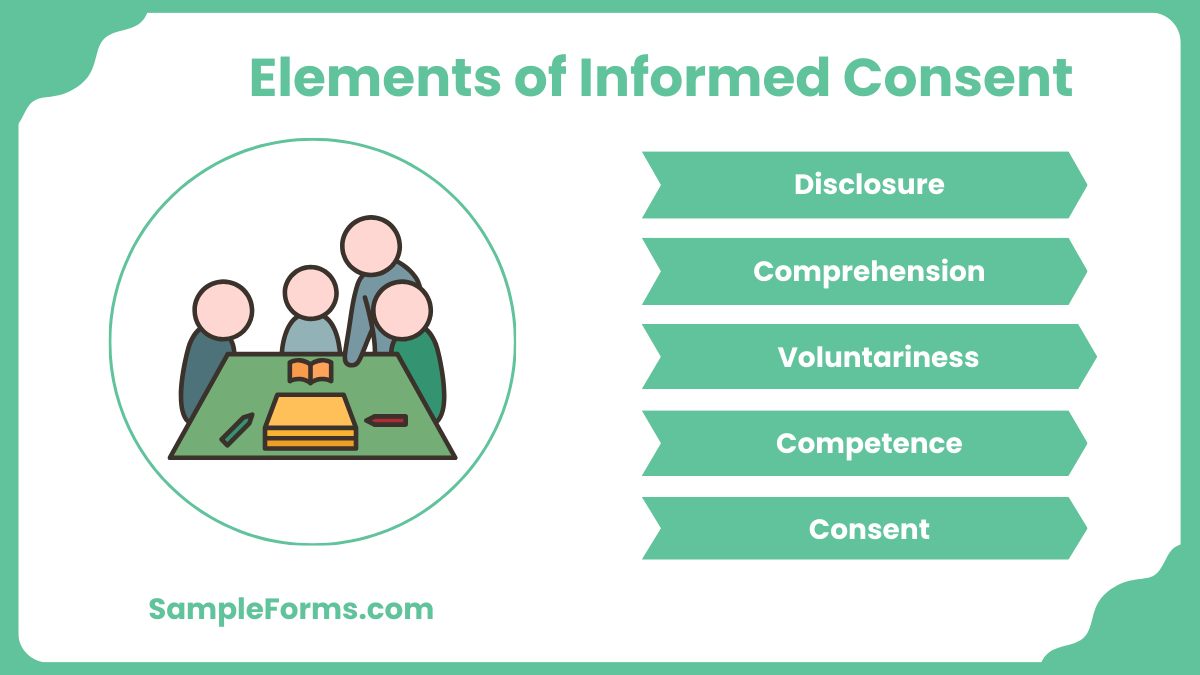
Elements To Include In Electronic Informed Consent Forms

What To Include In Hoa Electronic Informed Consent Forms Hoam What elements should you include in your form? it might be tempting to cut some information from your consent form to keep it simple or save space. however, the form must include all of the elements you need from the person filling it out and all the information you need to relay to them. Informed consent must be prospective, understandable, and not include exculpatory language. the consent process must also not create undue influence or coercion.
Free 12 Research Informed Consent Form Samples Pdf Ms Word Google Docs Outlines the key elements of informed consent, including the concise summary of key information, as mandated by both the revised common rule (45 cfr 46.116) and fda regulations (21 cfr 50.25). § 50.25 elements of informed consent. (a) basic elements of informed consent. in seeking informed consent, the following information shall be provided to each subject:. This article breaks down the key elements, documentation needs, and compliance strategies related to informed consent in clinical trials. what is informed consent under ich gcp?. Must contain all required elements of informed consent required by 45 cfr 46.116. must be written in a language understandable to the potential subject or subject’s legally authorized representative (lar) parent guardian and conveyed in a manner that minimizes coercion or undue influence.

Tips For Writing Informed Consent Forms This article breaks down the key elements, documentation needs, and compliance strategies related to informed consent in clinical trials. what is informed consent under ich gcp?. Must contain all required elements of informed consent required by 45 cfr 46.116. must be written in a language understandable to the potential subject or subject’s legally authorized representative (lar) parent guardian and conveyed in a manner that minimizes coercion or undue influence. Through an international, systematic scoping review, we identified and assessed all published findings on clinical e consent in the scholarly and grey literatures, including consents for telehealth encounters, procedures and health information exchanges. Informed consent form: what it is and what to include. whether you’re running a research study, getting a waiver, or going through an institutional review board, explore the guidelines and processes around informed consent form. The consent process starts with the initial presentation of a research activity to a prospective subject (including advertisements and notices), continues with a discussion and information exchange between the researcher and the prospective subject, and requires documenting that consent was obtained. Titute or add some electronic alternatives to the existing paper process. these include an electronic interactive participant education component instead of the paper icf, as well as an electronic signature that can be captured.
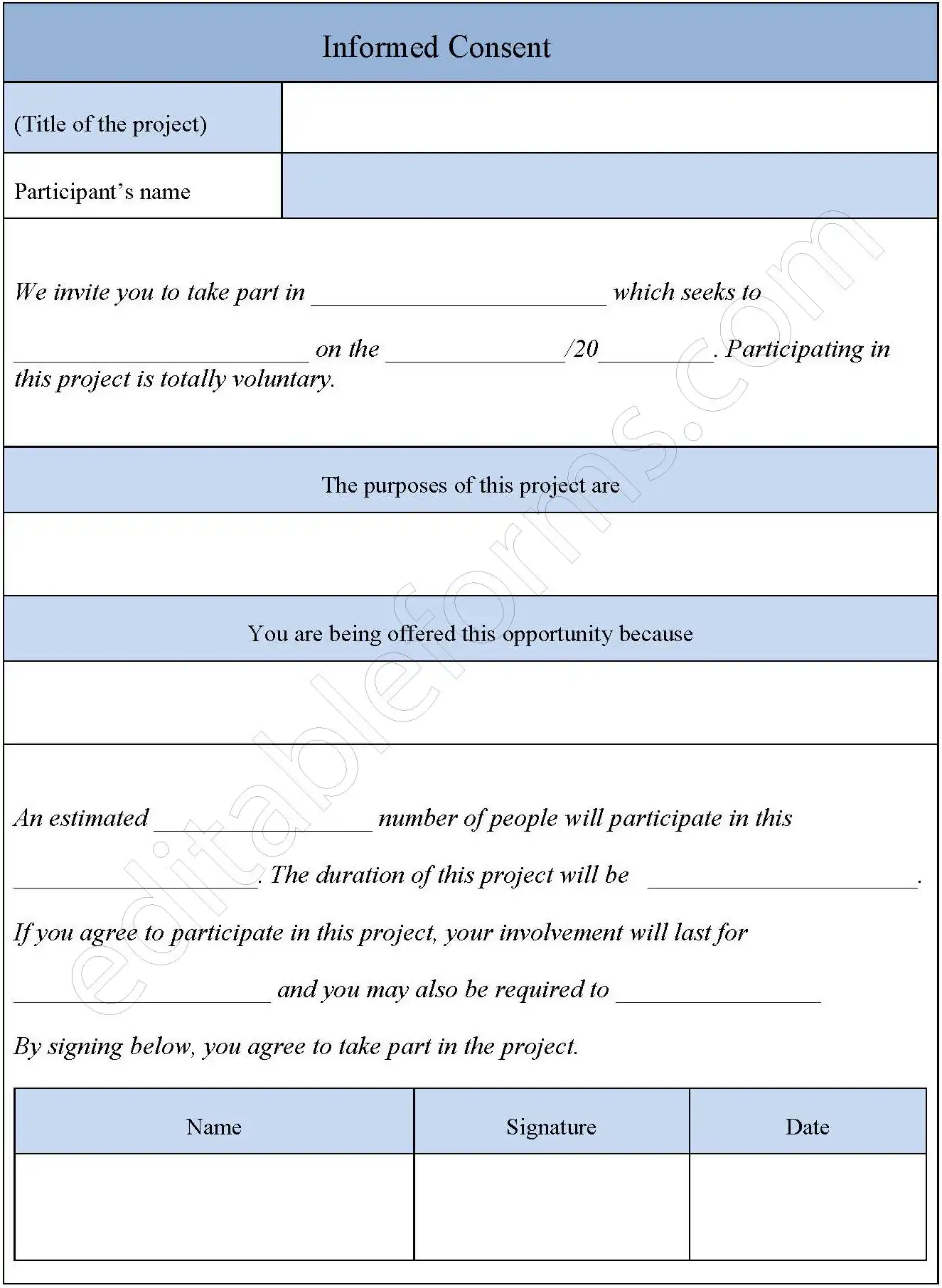
Informed Consent Form Template Editable Pdf Forms Through an international, systematic scoping review, we identified and assessed all published findings on clinical e consent in the scholarly and grey literatures, including consents for telehealth encounters, procedures and health information exchanges. Informed consent form: what it is and what to include. whether you’re running a research study, getting a waiver, or going through an institutional review board, explore the guidelines and processes around informed consent form. The consent process starts with the initial presentation of a research activity to a prospective subject (including advertisements and notices), continues with a discussion and information exchange between the researcher and the prospective subject, and requires documenting that consent was obtained. Titute or add some electronic alternatives to the existing paper process. these include an electronic interactive participant education component instead of the paper icf, as well as an electronic signature that can be captured.

9 Elements Of Informed Consent The consent process starts with the initial presentation of a research activity to a prospective subject (including advertisements and notices), continues with a discussion and information exchange between the researcher and the prospective subject, and requires documenting that consent was obtained. Titute or add some electronic alternatives to the existing paper process. these include an electronic interactive participant education component instead of the paper icf, as well as an electronic signature that can be captured.

Free 9 Sample Informed Consent Forms In Pdf Ms Word
Comments are closed.