Electronic Data Capture

Exploring The Progression Of Electronic Data Capture Edc In Clinical Mesin edc atau electronic data capture adalah perangkat yang digunakan oleh merchant untuk menerima pembayaran dari pelanggan menggunakan kartu kredit, kartu debit, kartu prepaid, dan qris. Learn about the history, features, and benefits of electronic data capture (edc), a computerized system for collecting clinical data in electronic format. edc replaces paper based data collection and is widely used by pharmaceutical and biotechnology companies in clinical trials.
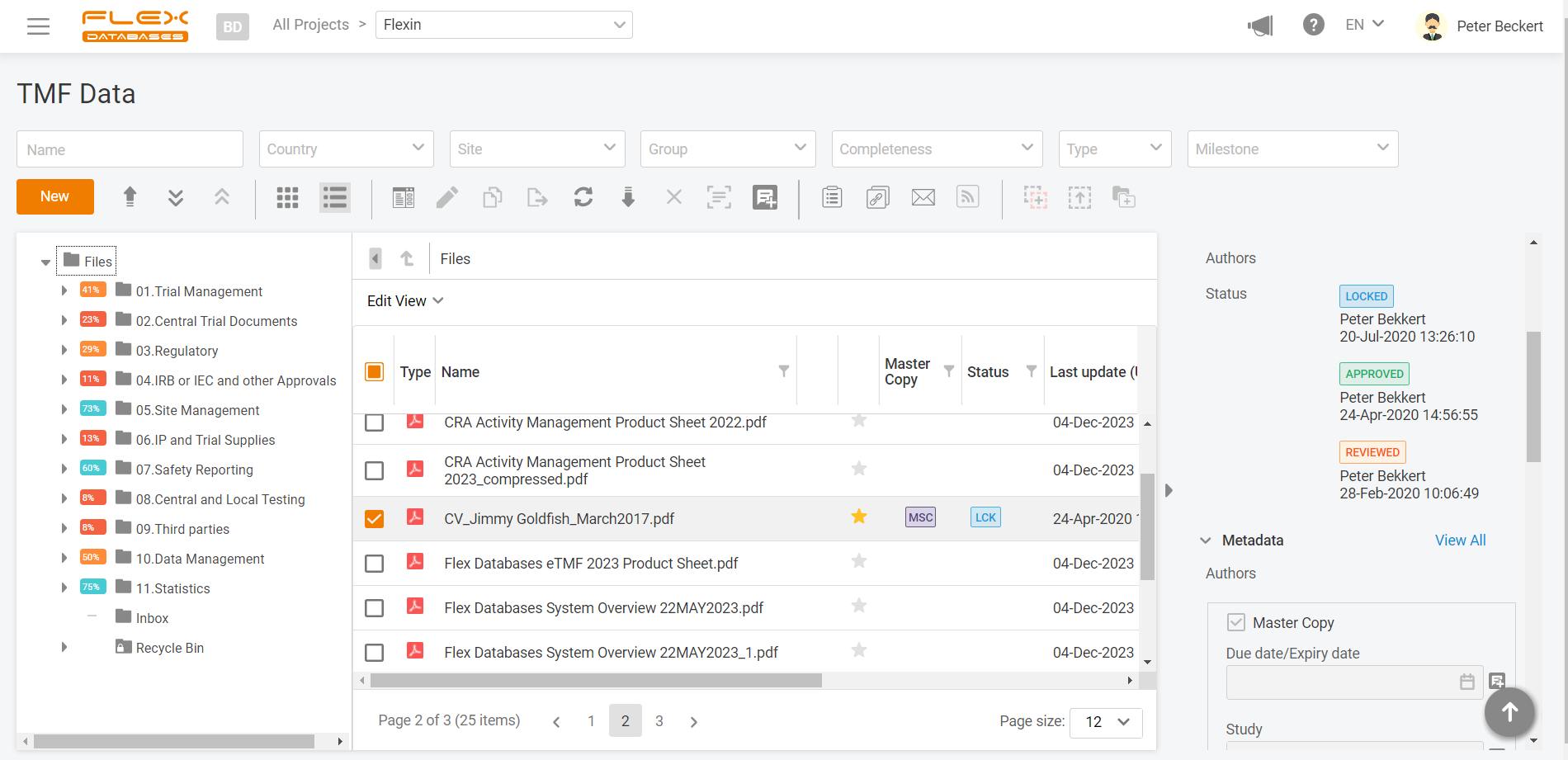
Electronic Data Capture Edc Clinical Trials Flex Databases Learn what edc is, how it works, and why it is important for medical device studies. compare edc with ecrf, ddc, esource, and other terms and concepts related to clinical data management. An electronic data capture (edc) system is a software platform designed to collect, manage, and store clinical trial data electronically. edc systems allow researchers to capture data directly from clinical trial sites, often in real time, replacing traditional paper based methods. What is electronic data capture? electronic data capture (edc) is an electronic system applied to clinical trials for capturing, managing, and storing patient data. instead of paper based forms, investigators enter clinical trial information directly into a secure, web based site. Learn about the leading electronic data capture (edc) systems for clinical trials and research in 2025, such as redcap, medidata rave, and di edc. compare their features, pricing, benefits, and drawbacks to choose the best one for your needs.
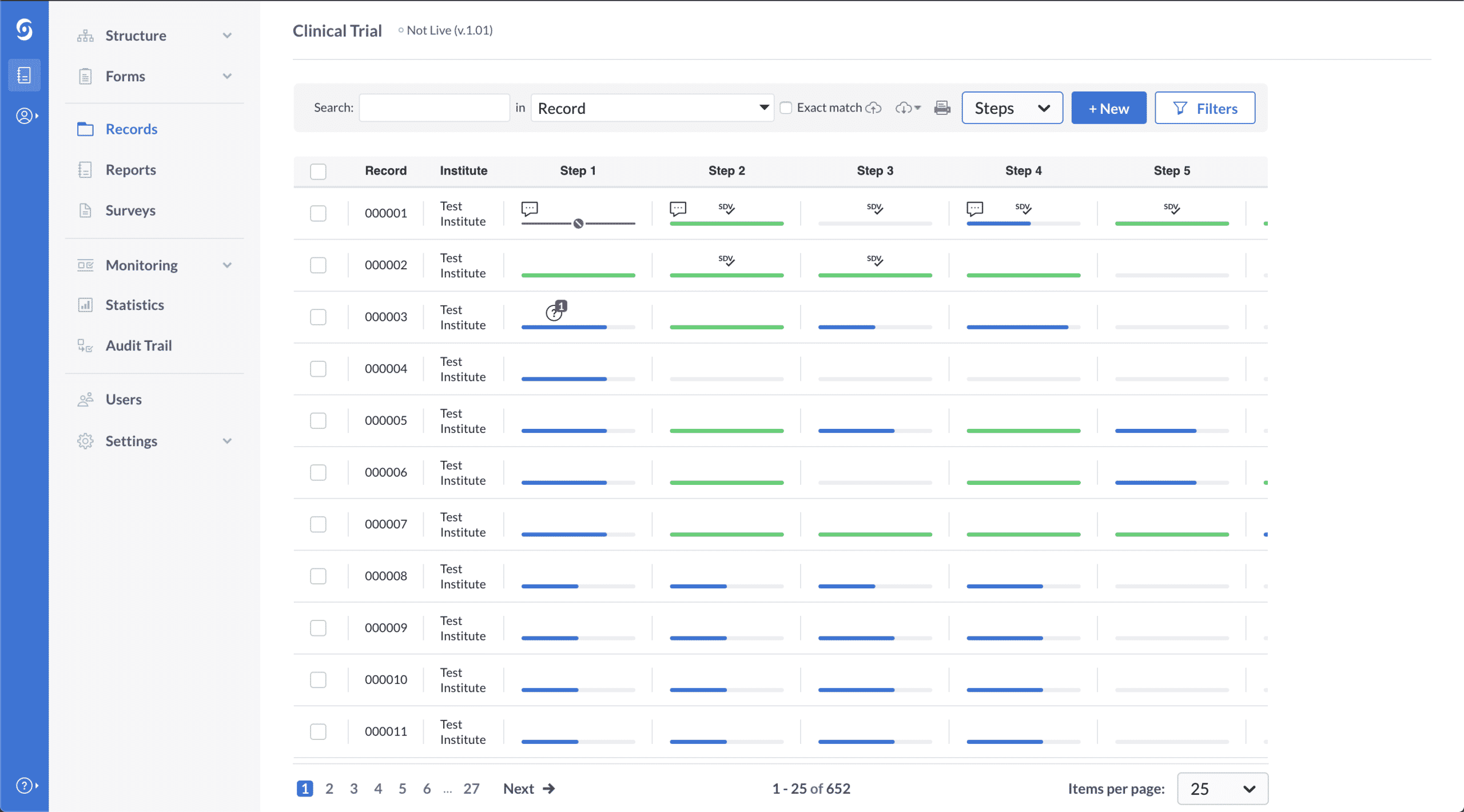
Electronic Data Capture Edc Platform For Clinical Trials What is electronic data capture? electronic data capture (edc) is an electronic system applied to clinical trials for capturing, managing, and storing patient data. instead of paper based forms, investigators enter clinical trial information directly into a secure, web based site. Learn about the leading electronic data capture (edc) systems for clinical trials and research in 2025, such as redcap, medidata rave, and di edc. compare their features, pricing, benefits, and drawbacks to choose the best one for your needs. To collect and manage structured data in digital format, researchers can use computer programs called electronic data capture (edc) systems [3]. there is a consensus that edc leads to improved data quality as well as cost and time efficiency compared with paper based methods. Electronic data capture (edc) software is a vital tool for efficiently collecting, managing, and analyzing clinical trial data. edc systems replace traditional paper based processes with secure, digital platforms that ensure data accuracy and accessibility. Electronic data capture (edc) is a technology used to collect data electronically—typically through specialized hardware or software designed to automatically capture and record information. edc is commonly used in fields such as healthcare, finance, retail, and clinical research. Electronic data capture (edc) is a sophisticated software system designed to collect, manage, and store clinical research data in electronic format.
Comments are closed.