Electrochemistry Cells Pdf

Electrochemistry Electrolytic Cells Pdf Electrochemistry Electrochemistry is the branch of the science that deals with the transformation of chemical energy into electrical energy and vice versa or it deals with the relationship between electrical and chemical energy produced in a redox reaction. The electrochemical potential (∆e0 cell), also known as cell potential, is the driving force for an electrochemical reaction and is related to the gibbs free energy change.

Electrochemistry Electrochemical Cells Pdf Electrochemistry Redox In brief, we are going to discuss electrochemical cells, which have the ability to produce electrical energy from chemical reactions, and also use electrical energy to create chemical reactions. Daniel cell is an example of a galvanic cell contains zinc immersed in zinc sulfate solution and copper in copper sulfate solution separated by a diaphragm or salt bridge (fig. 2.2). The goal of this chapter is to elucidate the fundamental characteristics and tech nological significance of electrochemical cells. An electrochemical (or voltaic) cell converts the energy change of a spontaneous reaction (chemical energy) into electrical energy. this electrical energy can do work and is proportional to the difference in potential between the two electrodes.

Electrochemistry Cells Pdf The goal of this chapter is to elucidate the fundamental characteristics and tech nological significance of electrochemical cells. An electrochemical (or voltaic) cell converts the energy change of a spontaneous reaction (chemical energy) into electrical energy. this electrical energy can do work and is proportional to the difference in potential between the two electrodes. Electrochemical cells allow measurement and control of a redox reaction. the reaction can be started and stopped by connecting or disconnecting the two electrodes. Electrochemistry is the study of chemical processes that involve electron movement. it is crucial in understanding reactions that convert chemical energy to electrical energy and vice versa. The figure above shows a set of electrochemical half cells that can be used to measure various voltages within galvanic cells. the cells shown are made of agar saturated with kcl solution so as to act as a salt bridge. We can construct innumerable number of galvanic cells on the pattern of daniell cell by taking combinations of different half cells. each half cell consists of a metallic electrode dipped into an electrolyte.
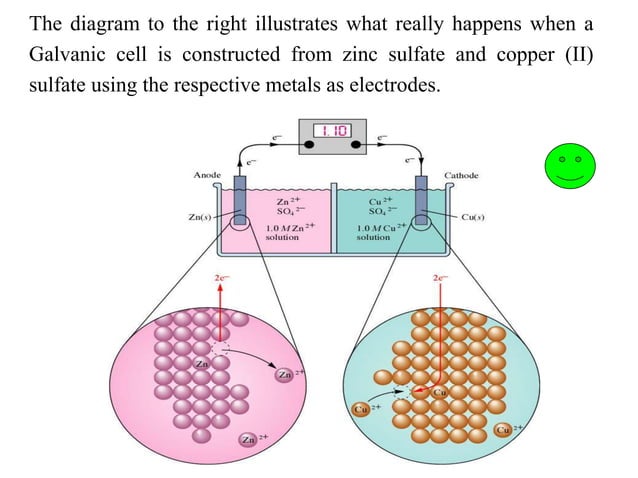
Electrochemistry Introduction Electrochemical Cells Ppt Electrochemical cells allow measurement and control of a redox reaction. the reaction can be started and stopped by connecting or disconnecting the two electrodes. Electrochemistry is the study of chemical processes that involve electron movement. it is crucial in understanding reactions that convert chemical energy to electrical energy and vice versa. The figure above shows a set of electrochemical half cells that can be used to measure various voltages within galvanic cells. the cells shown are made of agar saturated with kcl solution so as to act as a salt bridge. We can construct innumerable number of galvanic cells on the pattern of daniell cell by taking combinations of different half cells. each half cell consists of a metallic electrode dipped into an electrolyte.

Electrochemistry Pdf
Comments are closed.