Electrochemistry
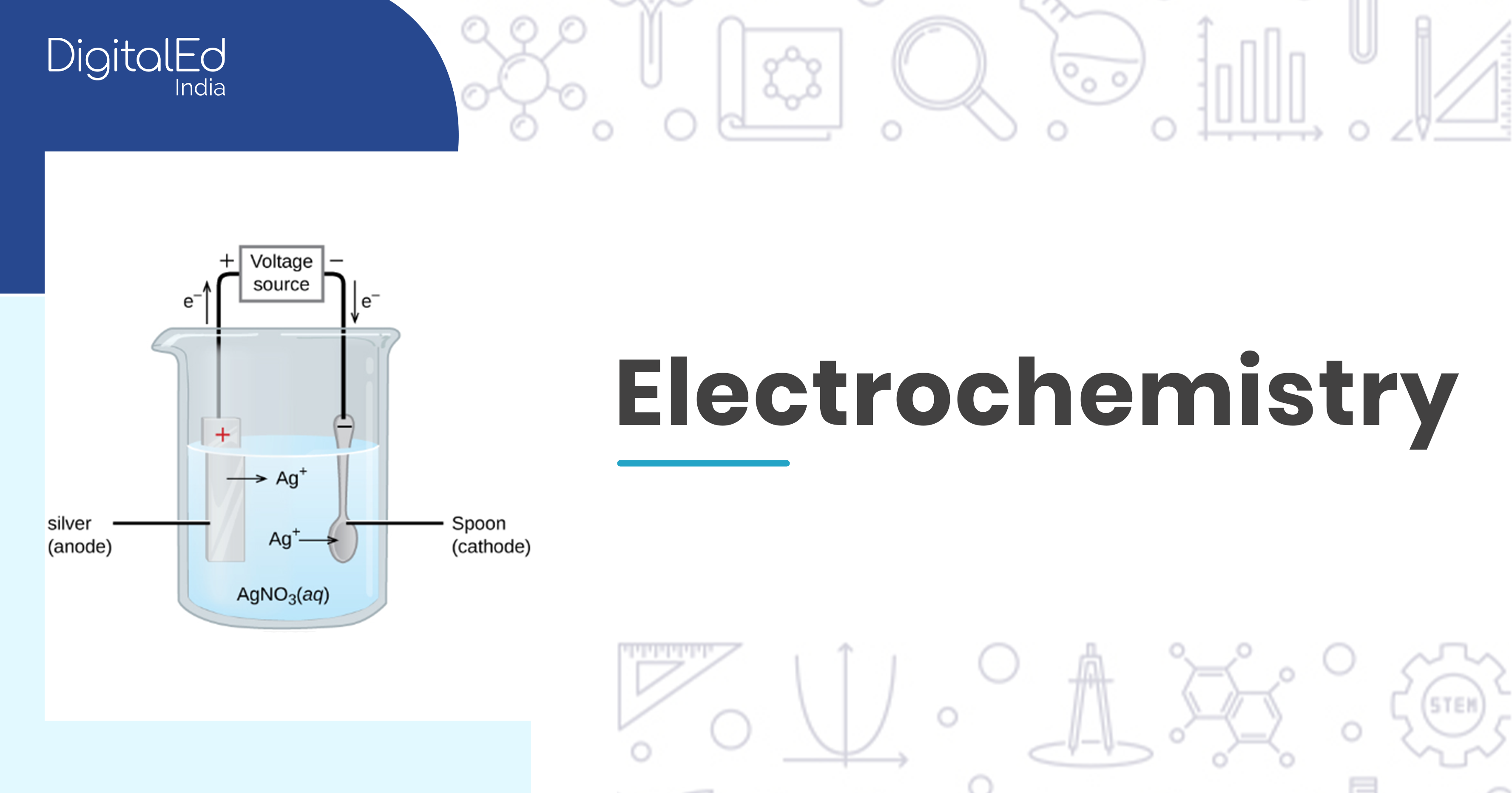
Electrochemistry Learn about the origins and development of electrochemistry, the branch of physical chemistry that studies the relationship between electricity and chemical change. explore the discoveries and inventions of pioneers such as galvani, volta, faraday, and daniell. Electrochemistry is the study of chemical processes that cause electrons to move. this movement of electrons is called electricity, which can be generated by movements of electrons from one element to another in a reaction known as an oxidation reduction ("redox") reaction.
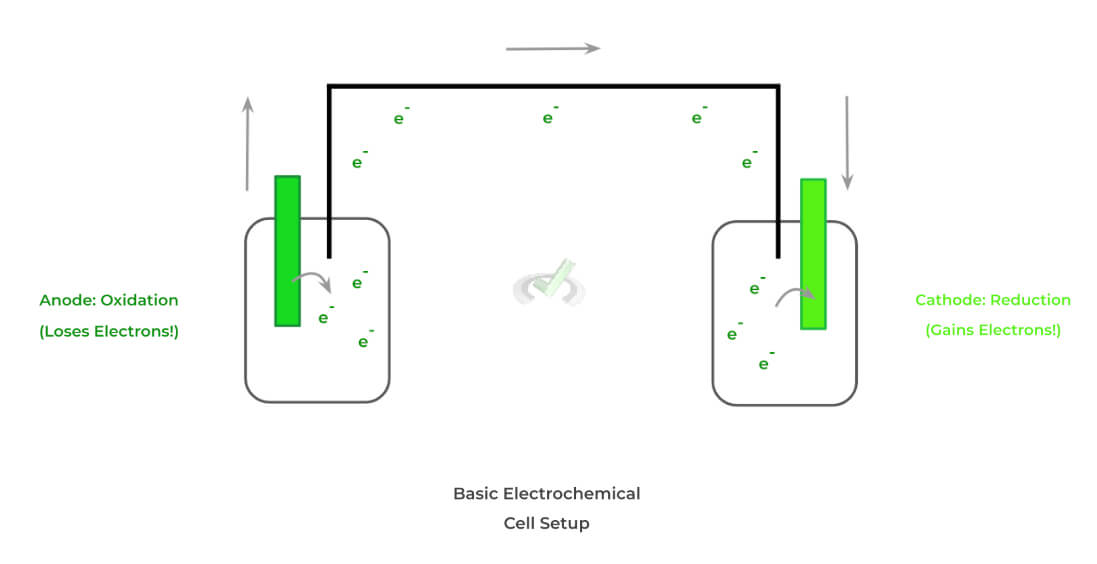
Electrochemistry On The Mcat Medlife Mastery Electrochemistry is the study of electron movement in an oxidation or reduction reaction at a polarized electrode surface. each analyte is oxidized or reduced at a specific potential and the current measured is proportional to concentration. Electrochemistry, branch of chemistry concerned with the relation between electricity and chemical change. many spontaneously occurring chemical reactions liberate electrical energy, and some of these reactions are used in batteries and fuel cells to produce electric power. Learn about electrochemistry, the study of how electrical energy and chemical changes interact. explore electrolysis, galvanic cells, redox reactions, and applications of electrochemistry in various fields. This site contains popular science style articles describing many aspects of electrochemistry, electroanalytical chemistry, electrochemical engineering, and electrochemical technology, written by international experts in the field.
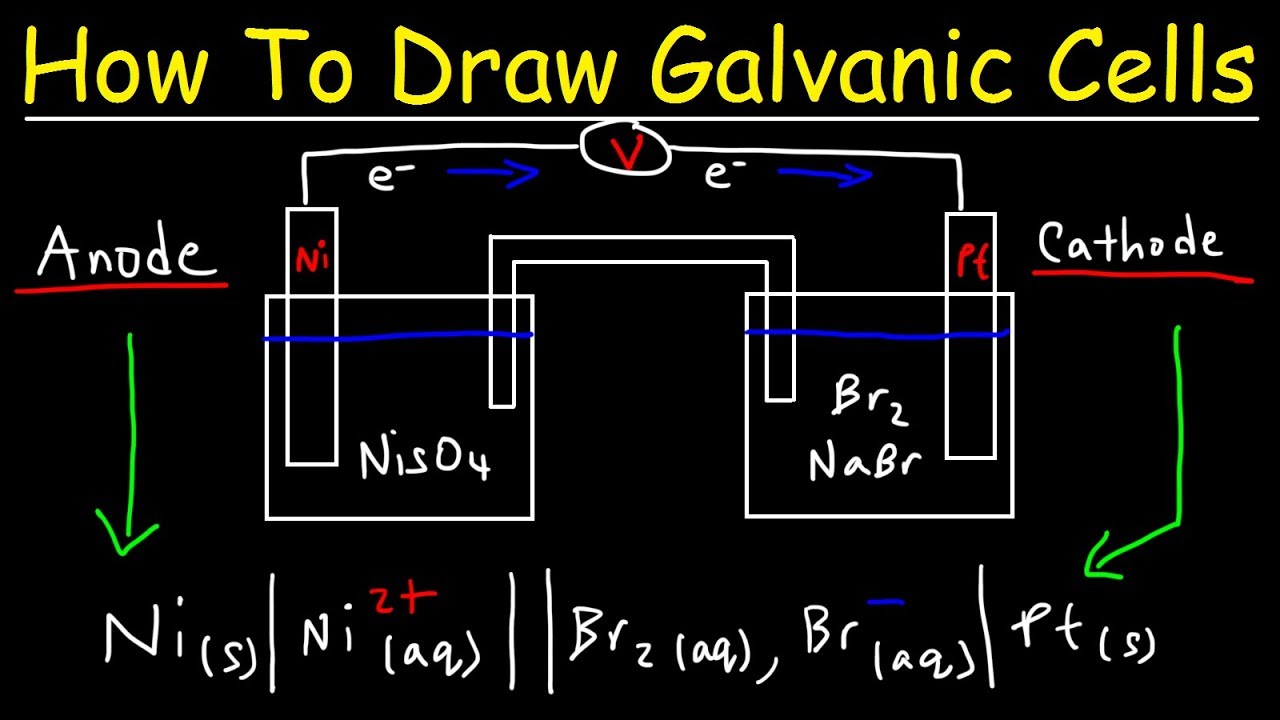
Electrochemistry And Solutions Concept Map Chemistry Learn about electrochemistry, the study of how electrical energy and chemical changes interact. explore electrolysis, galvanic cells, redox reactions, and applications of electrochemistry in various fields. This site contains popular science style articles describing many aspects of electrochemistry, electroanalytical chemistry, electrochemical engineering, and electrochemical technology, written by international experts in the field. The chapter “fundamentals of electrochemistry” provides a comprehensive overview of the core principles governing electrochemical processes. it begins with an introduction to electrochemical processes, laying the groundwork for understanding the underlying mechanisms. In the two centuries since, the field of electrochemistry has evolved to yield significant insights on the fundamental aspects of redox chemistry as well as a wealth of technologies ranging from industrial scale metallurgical processes to robust, rechargeable batteries for electric vehicles (figure 1). Learn about electrochemistry, the branch of chemistry that deals with chemical reactions that produce electricity and vice versa. explore the basics of oxidation reduction reactions, galvanic cells, batteries, corrosion, and electrolysis. Learn the fundamentals of electrochemistry, including redox reactions, galvanic cells, and how to use the nernst equation. explore the applications of electrochemistry in batteries, fuel cells, and electrolysis.

Ppt Electrochemistry Mae 212 Powerpoint Presentation Free Download The chapter “fundamentals of electrochemistry” provides a comprehensive overview of the core principles governing electrochemical processes. it begins with an introduction to electrochemical processes, laying the groundwork for understanding the underlying mechanisms. In the two centuries since, the field of electrochemistry has evolved to yield significant insights on the fundamental aspects of redox chemistry as well as a wealth of technologies ranging from industrial scale metallurgical processes to robust, rechargeable batteries for electric vehicles (figure 1). Learn about electrochemistry, the branch of chemistry that deals with chemical reactions that produce electricity and vice versa. explore the basics of oxidation reduction reactions, galvanic cells, batteries, corrosion, and electrolysis. Learn the fundamentals of electrochemistry, including redox reactions, galvanic cells, and how to use the nernst equation. explore the applications of electrochemistry in batteries, fuel cells, and electrolysis.
Comments are closed.