Electric Fields Problems Pdf Proton Atomic Nucleus

Physics Ii Problems Pdf Pdf Atomic Nucleus Proton Electric fields problems free download as pdf file (.pdf), text file (.txt) or read online for free. Selected problems for chapter 23: electric field ypically separated by a distance of 2 × 10–15 m. the electric repulsion force between the protons is huge, but the attractive nuclear force is even stronger and keeps the nucleus from bursting apart. what is the magnitude of the electric force ee.

Understanding Electric Fields Problems Solutions Course Hero Assume that the nucleus is a sphere with radius 7.26 fm and with the charge of the protons uniformly spread through the sphere. at the nucleus surface what are (a) the magnitude and (b) direction (radially inward or outward) of the electric eld produced by the protons?. Calculate the electric field strength at a distance of 2.0 10 10 m from the proton. calculate the electrical potential at a distance of 2.0 10 10 m from the proton. The atom consists of a positively charged nucleus surrounded by negatively charged electrons. the nucleus consists of protons, which have positive charge, and neutrons, which have no charge. the charge of a proton is the same in magnitude but opposite in sign to that of an electron. In the borh’s hydrogen model, the electron is imagined to move in a circular orbit about a stationary proton. the force responsible for the electron circular motion is the electric force between the electron and the proton.

Chapter 18 Electric Forces And Electric Fields The atom consists of a positively charged nucleus surrounded by negatively charged electrons. the nucleus consists of protons, which have positive charge, and neutrons, which have no charge. the charge of a proton is the same in magnitude but opposite in sign to that of an electron. In the borh’s hydrogen model, the electron is imagined to move in a circular orbit about a stationary proton. the force responsible for the electron circular motion is the electric force between the electron and the proton. The proton in the nucleus of the hydrogen atom attracts the electron that orbits it with electric force. relative to this force, how does the electron attracts the proton?. If a proton is released from rest in an electric field, will it move in the direction of increasing or decreasing potential? also answer this question for an electron and a neutron. This volume on atomic, nuclear and particle physics which contains 483 problems is divided into four parts: atomic and molecular physics (142), nuclear physics (120), particle physics (90), experimental methods and miscellaneous topics (131). Orbital and spin angular momenta of protons produce magnetic field within the nucleus. this field can be described in terms of resultant magnetic dipole moment located at the centre of the nucleus.
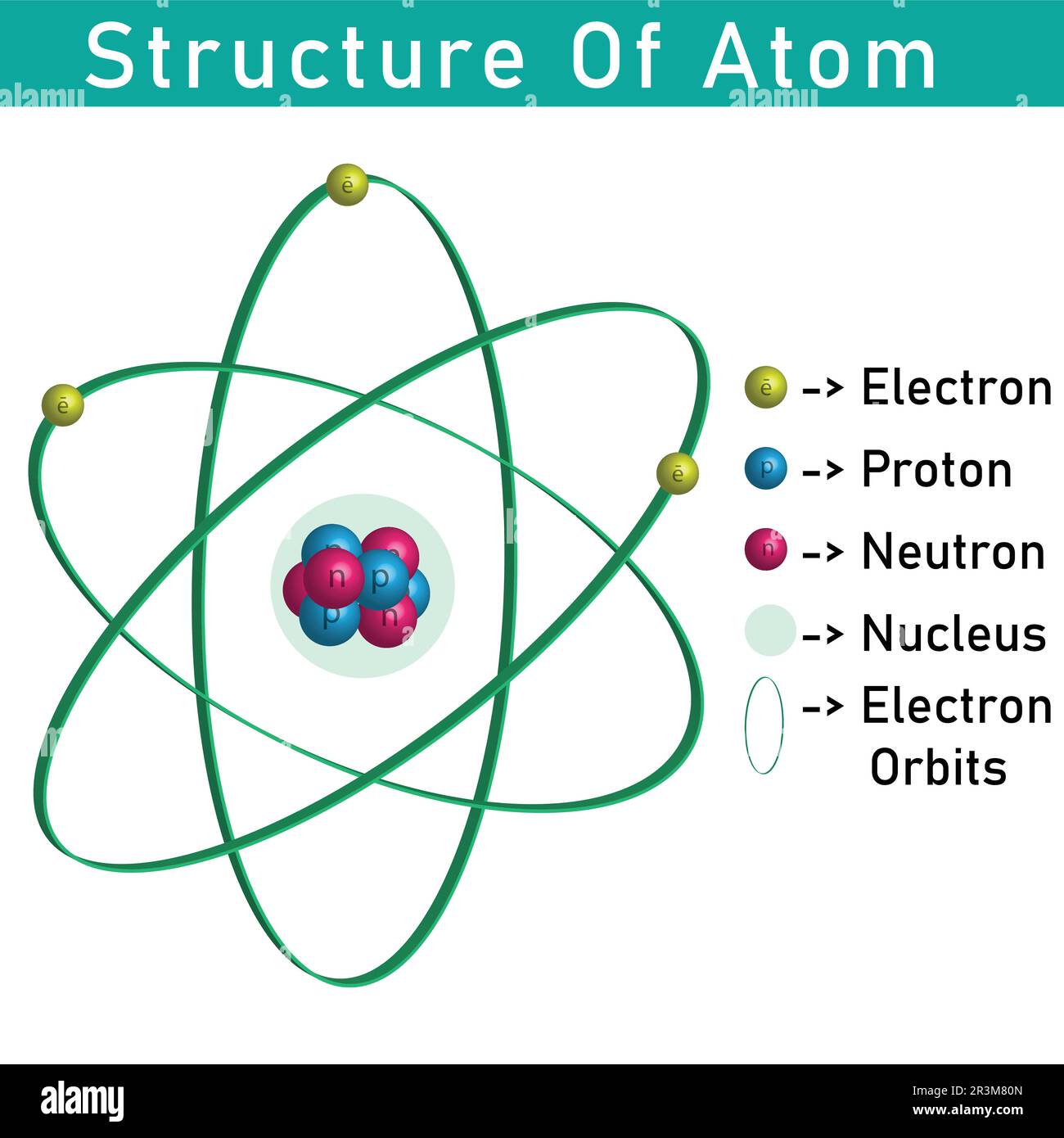
Projeto Proton Neutron Eletron 1 2 The Nuclear Atom Chemistry The proton in the nucleus of the hydrogen atom attracts the electron that orbits it with electric force. relative to this force, how does the electron attracts the proton?. If a proton is released from rest in an electric field, will it move in the direction of increasing or decreasing potential? also answer this question for an electron and a neutron. This volume on atomic, nuclear and particle physics which contains 483 problems is divided into four parts: atomic and molecular physics (142), nuclear physics (120), particle physics (90), experimental methods and miscellaneous topics (131). Orbital and spin angular momenta of protons produce magnetic field within the nucleus. this field can be described in terms of resultant magnetic dipole moment located at the centre of the nucleus.
Comments are closed.