Distillation Of Water And Ethanol

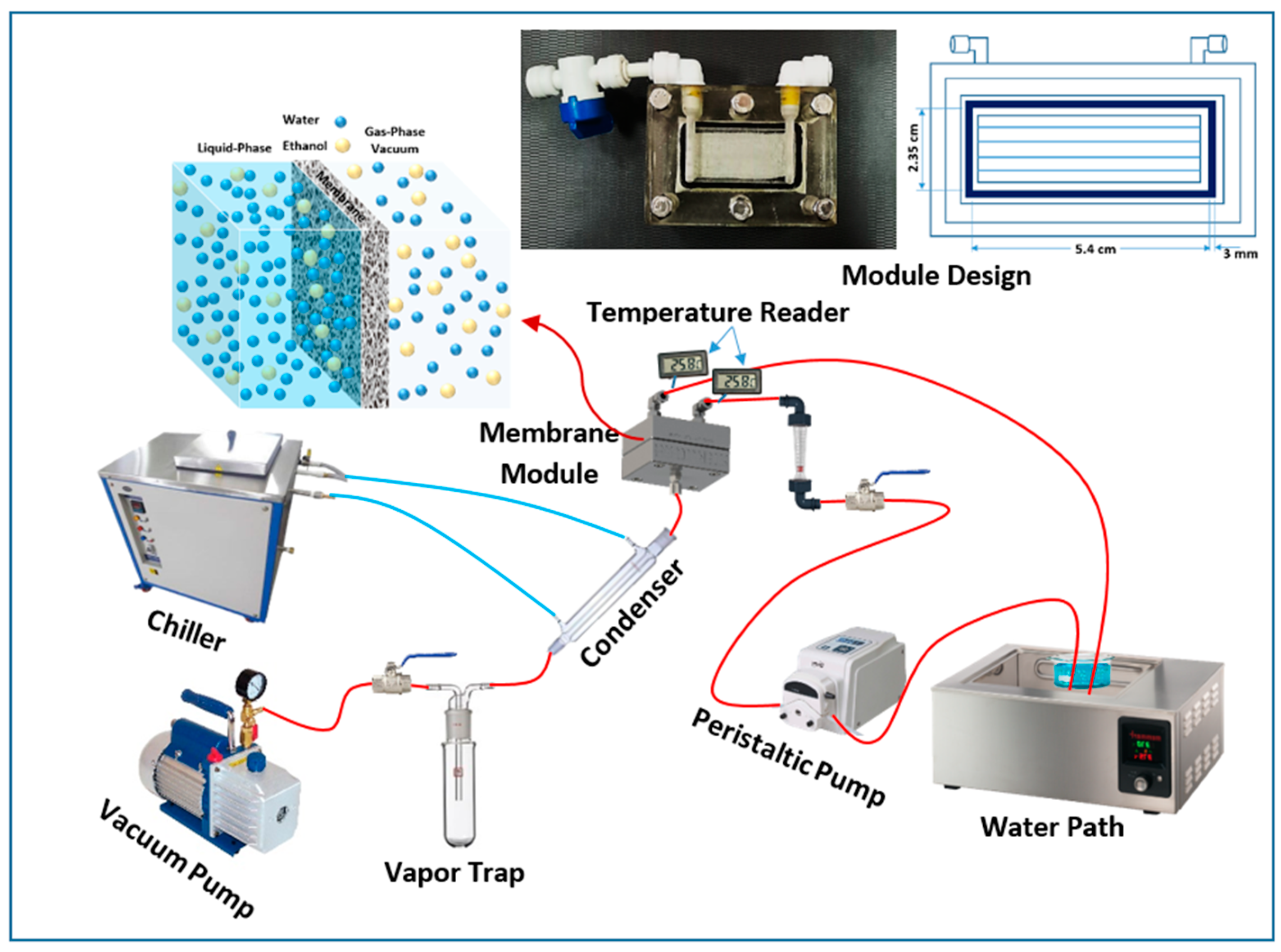
Amasci Net Ethanol Purification Method 1 The vacuum membrane distillation (vmd) process was applied to separate ethanol from a simulated ethanol–water solution using a commercial polytetrafluoroethylene (ptfe) membrane. This work aims to design an extractive distillation process that is intensified by an il for the separation of ethanol water mixture with a high ethanol purity of 99.9%.

Ethanol Distillation Ethanol Distillation Process Eureka Patsnap Extractive distillation is a specialized distillation process that involves adding an additional component (extractive agent or solvent entrainer) to the mixture in order to alter the relative. It is impossible to get pure ethanol by distilling any mixture of ethanol and water containing less than 95.6% of ethanol. this particular mixture of ethanol and water boils as if it were a pure liquid. it has a constant boiling point, and the vapor composition is exactly the same as the liquid. An equimolar binary mixture of ethanol and water is to be separated in a still pot that initially contains 0.575 kmol of the mixture. you can set the operating pressure, p, of this batch distillation, where p can vary from 700 to 900 mmhg so that the ideal gas assumption holds. Distillation is a thermal separation process that exploits differences in boiling points to separate components in a liquid mixture. in the case of ethanol and water, ethanol has a lower boiling point (78.37°c) compared to water (100°c), which makes the separation feasible.
Vacuum Distillation Ethanol Water At Scott Gerber Blog An equimolar binary mixture of ethanol and water is to be separated in a still pot that initially contains 0.575 kmol of the mixture. you can set the operating pressure, p, of this batch distillation, where p can vary from 700 to 900 mmhg so that the ideal gas assumption holds. Distillation is a thermal separation process that exploits differences in boiling points to separate components in a liquid mixture. in the case of ethanol and water, ethanol has a lower boiling point (78.37°c) compared to water (100°c), which makes the separation feasible. Explore the science and mechanics of ethanol distillation, revealing how boiling points separate liquids and why the azeotrope prevents maximum purity. Learn how to separate ethanol and water. we detail methods for overcoming the azeotrope, utilizing distillation, desiccation, and chemical entrainers. Ethanol (bp 78 °c) has a lower boiling point than water (bp 100 °c). because ethanol boils before water, during a distillation, ethanol can be distilled and collected while the water remains in the distilling pot. Learning objectives perform a simple distillation of a 20% ethanol water mixture. perform a fractional distillation of a 20% ethanol water mixture. compare the efficacy of simple distillation vs. fractional distillation.

Vacuum Distillation Ethanol Water At Scott Gerber Blog Explore the science and mechanics of ethanol distillation, revealing how boiling points separate liquids and why the azeotrope prevents maximum purity. Learn how to separate ethanol and water. we detail methods for overcoming the azeotrope, utilizing distillation, desiccation, and chemical entrainers. Ethanol (bp 78 °c) has a lower boiling point than water (bp 100 °c). because ethanol boils before water, during a distillation, ethanol can be distilled and collected while the water remains in the distilling pot. Learning objectives perform a simple distillation of a 20% ethanol water mixture. perform a fractional distillation of a 20% ethanol water mixture. compare the efficacy of simple distillation vs. fractional distillation.

Ethanol Distillation Ethanol (bp 78 °c) has a lower boiling point than water (bp 100 °c). because ethanol boils before water, during a distillation, ethanol can be distilled and collected while the water remains in the distilling pot. Learning objectives perform a simple distillation of a 20% ethanol water mixture. perform a fractional distillation of a 20% ethanol water mixture. compare the efficacy of simple distillation vs. fractional distillation.
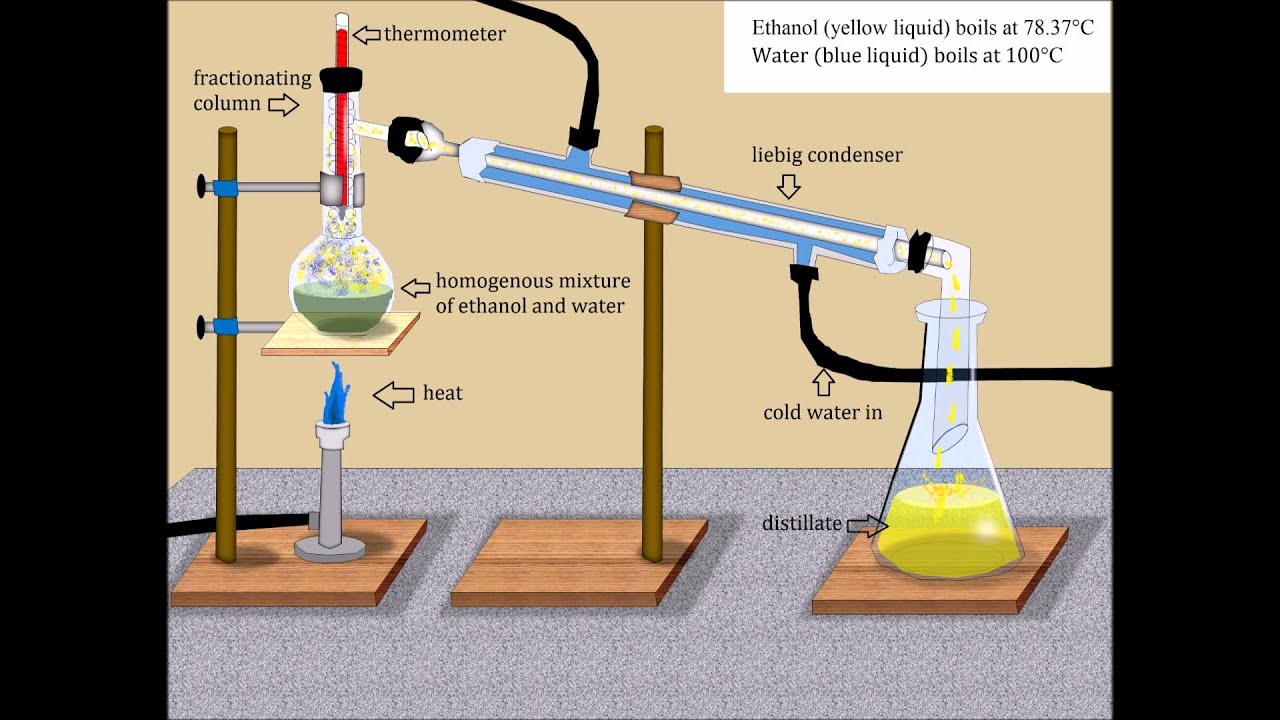
Ethanol Distillation
Comments are closed.