Distillation Alcohol Oxidation

10 2 C Alcohol Reactions Combustion Oxidation Distillation Reflux Learn about oxidation of alcohols for your a level chemistry exam. find information on primary and secondary alcohols and distillation vs reflux. A very commonly example is the oxidation of an alcohol to a ketone or aldehyde. notice that during this process the carbon atom loses a hydrogen and gains a bond to oxygen.
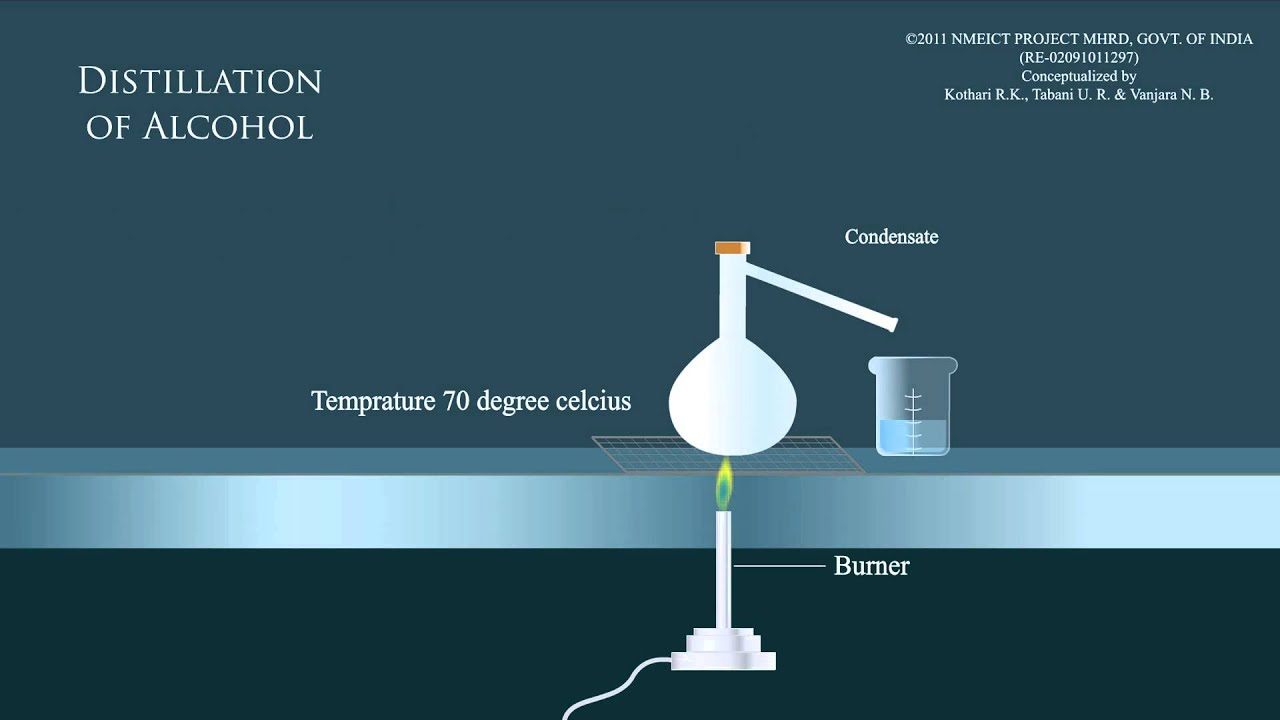
Alcohol Distillation Primary alcohols can be oxidised to an aldehyde, then to a carboxylic acid. to isolate the aldehyde, the products must be distilled from the reaction mixture. if a carboxylic acid is desired, the mixture must be heated under reflux conditions. secondary alcohols can only be oxidised to form ketones. As an example of the oxidation process consider the oxidation of the primary alcohol ethanol to the aldehyde ethanal. the apparatus set up to carry out this oxidation reaction is shown below, the set up is simple distillation. As the aldehyde forms, it evaporates and is collected by distillation, removing it from the reaction mixture and preventing further oxidation. The aldehyde, since its boiling point is lower than the alcohol, can be separated out through distillation. refluxing with excess [o] leads to further oxidation and the formation of a carboxylic acid.
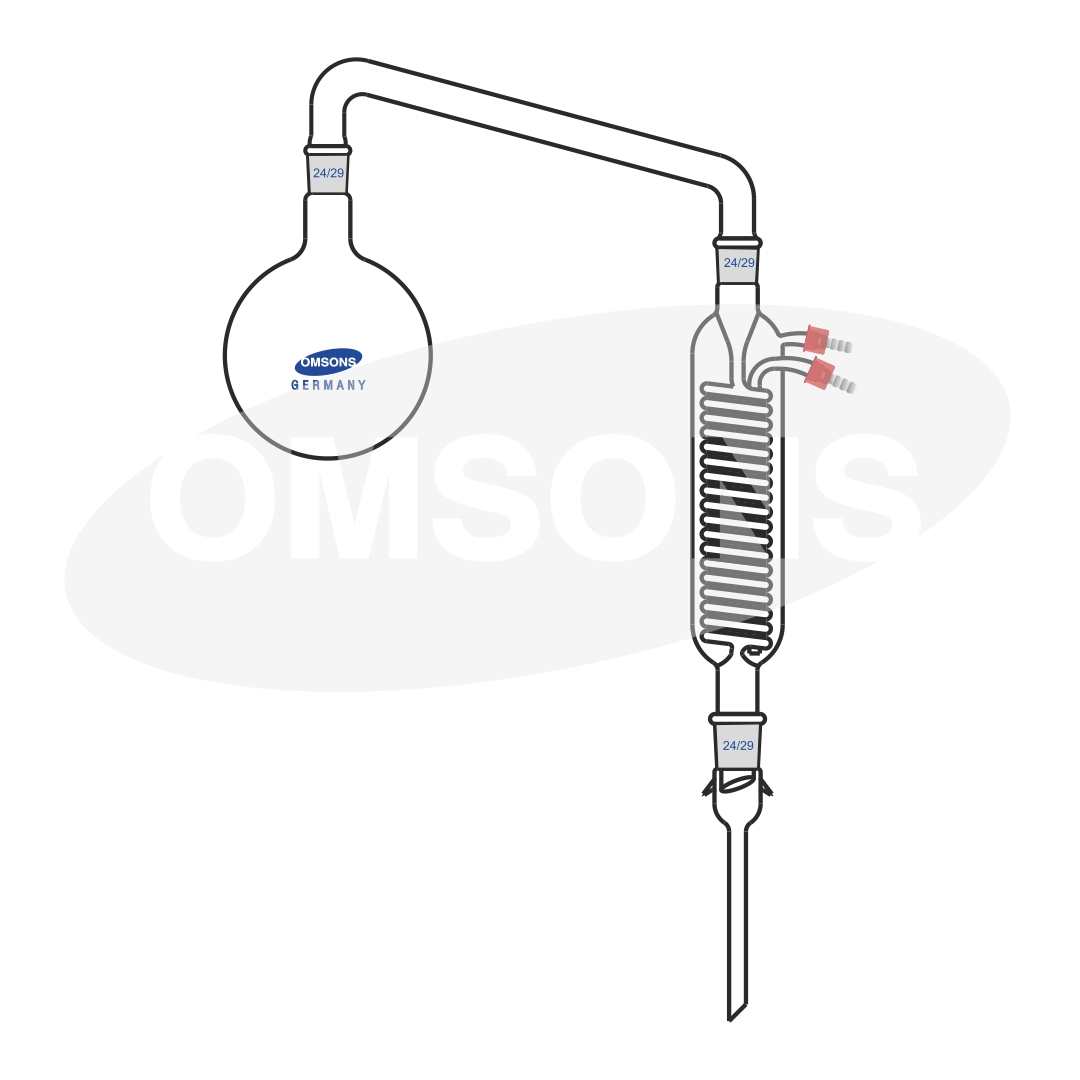
Alcohol Distillation As the aldehyde forms, it evaporates and is collected by distillation, removing it from the reaction mixture and preventing further oxidation. The aldehyde, since its boiling point is lower than the alcohol, can be separated out through distillation. refluxing with excess [o] leads to further oxidation and the formation of a carboxylic acid. In the case of the formation of carboxylic acids, the alcohol is first oxidized to an aldehyde, which is then oxidized further to the acid. an aldehyde is obtained if an excess amount of the alcohol is used, and the aldehyde is distilled off as soon as it forms. The mechanism of alcohol oxidation typically involves the reduction of the oxidizing agent and the formation of a carbon oxygen double bond. in this article, we look into what alcohol is, the types of alcohols, the mechanism of oxidation of alcohols, uses of alcohols, etc. In the case of the formation of carboxylic acids, the alcohol is first oxidised to an aldehyde which is then oxidised further to the acid. you get an aldehyde if you use an excess of the alcohol, and distil off the aldehyde as soon as it forms. Primary alcohols can be oxidized to form an aldehyde. the aldehyde will have a lower boiling point than the alcohol and can be distilled off. if however the alcohol is heated under reflux, the aldehyde does not evaporate off and stays in the reaction vessel, which causes it to become further oxidized to form a carboxylic acid.

Alcohol Oxidation Reactor With Ethanol Distillation Column In the case of the formation of carboxylic acids, the alcohol is first oxidized to an aldehyde, which is then oxidized further to the acid. an aldehyde is obtained if an excess amount of the alcohol is used, and the aldehyde is distilled off as soon as it forms. The mechanism of alcohol oxidation typically involves the reduction of the oxidizing agent and the formation of a carbon oxygen double bond. in this article, we look into what alcohol is, the types of alcohols, the mechanism of oxidation of alcohols, uses of alcohols, etc. In the case of the formation of carboxylic acids, the alcohol is first oxidised to an aldehyde which is then oxidised further to the acid. you get an aldehyde if you use an excess of the alcohol, and distil off the aldehyde as soon as it forms. Primary alcohols can be oxidized to form an aldehyde. the aldehyde will have a lower boiling point than the alcohol and can be distilled off. if however the alcohol is heated under reflux, the aldehyde does not evaporate off and stays in the reaction vessel, which causes it to become further oxidized to form a carboxylic acid.
Comments are closed.