Difference Between Solid Liquid And Gas

What Is The Difference Between A Solid Liquid And Gas Kidpid Learn the meaning and properties of solid, liquid and gas, the three fundamental states of matter. compare their shape, volume, energy, compression, molecular arrangement, fluidity and speed of sound with a table and examples. Learn the difference between solid, liquid and gas states of matter based on their intermolecular forces, shape, volume and compressibility. find examples, videos and faqs on this topic.

Difference Between Solid Liquid And Gas Geeksforgeeks There are three basic states of matter that include, solid, liquid, and gas. these three states of matter have various differences and learning the differences between solid, liquid, and gas are very important for the understanding of matter. Discover how solids, liquids and gases differ in chemistry. learn how particle behavior defines each state and see real‑world examples. Three states of matter exist: solid, liquid, and gas. solids have a definite shape and volume. liquids have a definite volume, but take the shape of the container. gases have no definite shape or …. Find out what particle arrangements and movements are in solids, liquids, and gases in this bbc bitesize ks3 physics guide.
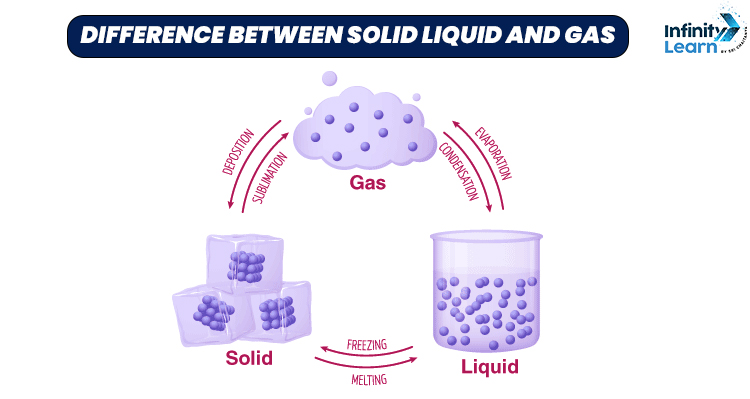
Difference Between Solid Liquid And Gas Properties And States Three states of matter exist: solid, liquid, and gas. solids have a definite shape and volume. liquids have a definite volume, but take the shape of the container. gases have no definite shape or …. Find out what particle arrangements and movements are in solids, liquids, and gases in this bbc bitesize ks3 physics guide. Understand the key differences between solids, liquids, and gases with their properties and examples. learn how their shape, volume, and molecular arrangement vary. Understanding these differences is important in physics because it helps us explain natural phenomena like melting, boiling, evaporation, expansion, and compression. below is a clear explanation of how solids, liquids, and gases differ based on particle arrangement, movement, shape, and volume. Learn the characteristics and properties of solids, liquids, and gases with examples and a tabular comparison. find out how intermolecular forces, volume, density, diffusion, and kinetic energy affect the states of matter. Liquids maintain a fixed volume but lack a definite shape. a liquid will flow to take on the exact shape of the portion of the container it occupies. the volume of the liquid remains relatively constant. gases are characterized by having neither a definite shape nor a fixed volume.

Difference Between Solid Liquid Gas In Table Form Teachoo Understand the key differences between solids, liquids, and gases with their properties and examples. learn how their shape, volume, and molecular arrangement vary. Understanding these differences is important in physics because it helps us explain natural phenomena like melting, boiling, evaporation, expansion, and compression. below is a clear explanation of how solids, liquids, and gases differ based on particle arrangement, movement, shape, and volume. Learn the characteristics and properties of solids, liquids, and gases with examples and a tabular comparison. find out how intermolecular forces, volume, density, diffusion, and kinetic energy affect the states of matter. Liquids maintain a fixed volume but lack a definite shape. a liquid will flow to take on the exact shape of the portion of the container it occupies. the volume of the liquid remains relatively constant. gases are characterized by having neither a definite shape nor a fixed volume.
Comments are closed.