Difference Between S And P Block Elements Definition Characteristic

Notes S P Block Elements Premed Pk Pdf Sodium Chlorine What is the difference between s and p block elements? all s block elements are metals; most of the p block elements are nonmetals, others are metalloids. They are characterized by their electronic configurations, where s block elements have their outermost electrons in s orbitals, while p block elements have their outermost electrons in p orbitals.

Ch 13 S And P Block Elements Pdf Comparative study of s and p block elements the document outlines a comparative study of s and p block elements in chemistry, detailing their definitions, properties, and periodic trends. The defining characteristic that separates s block and p block elements lies in their electronic configuration, specifically the filling of their outermost electron shells. While s block elements have their outermost electrons in the s orbital and are highly reactive metals, p block elements have their outermost electrons in the p orbital and exhibit a wide range of reactivity and physical properties. The difference in inner core of the elements greatly influences many physical as well as chemical properties of p block elements. metal, non metal and metalloid are present in p block elements.
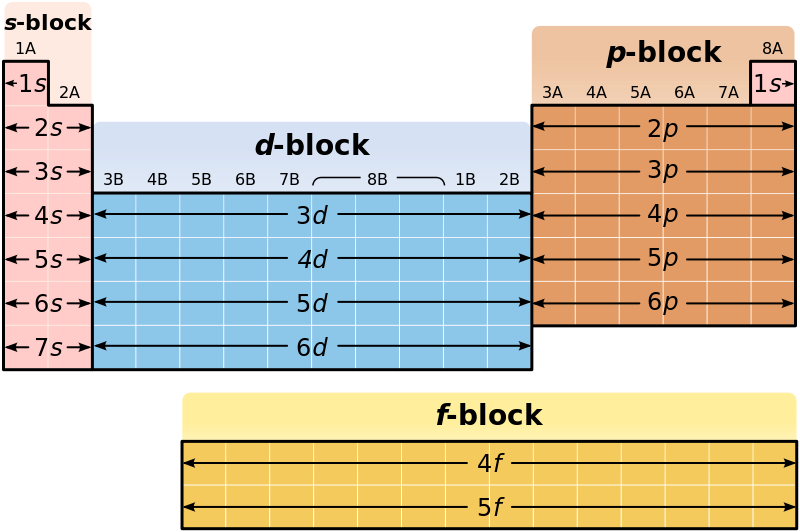
Difference Between S And P Block Elements Definition Characteristic While s block elements have their outermost electrons in the s orbital and are highly reactive metals, p block elements have their outermost electrons in the p orbital and exhibit a wide range of reactivity and physical properties. The difference in inner core of the elements greatly influences many physical as well as chemical properties of p block elements. metal, non metal and metalloid are present in p block elements. S block elements show only one oxidation state. p block elements are capable of showing more than one oxidation state. for example, oxidation states of oxygen in different oxides,. This comparison covers their position in the periodic table, oxidation states, reactivity, metallic character, and special properties in a clear tabular format along with faqs for quick revision. In terms of the electron configuration, a group consists of a series of elements which have the same outermost electronic configurations but they have different total numbers of electrons. Periodic table blocks are sets of elements grouped by their valence electron orbitals. the four block names are s block, p block, d block, and f block. should a new element be discovered, it will be in g block. each block indicates which electron sublevel is in the process of being filled.

Difference Between S And P Block Elements Definition Characteristic S block elements show only one oxidation state. p block elements are capable of showing more than one oxidation state. for example, oxidation states of oxygen in different oxides,. This comparison covers their position in the periodic table, oxidation states, reactivity, metallic character, and special properties in a clear tabular format along with faqs for quick revision. In terms of the electron configuration, a group consists of a series of elements which have the same outermost electronic configurations but they have different total numbers of electrons. Periodic table blocks are sets of elements grouped by their valence electron orbitals. the four block names are s block, p block, d block, and f block. should a new element be discovered, it will be in g block. each block indicates which electron sublevel is in the process of being filled.
Difference Between S And P Block Elements Definition Characteristic In terms of the electron configuration, a group consists of a series of elements which have the same outermost electronic configurations but they have different total numbers of electrons. Periodic table blocks are sets of elements grouped by their valence electron orbitals. the four block names are s block, p block, d block, and f block. should a new element be discovered, it will be in g block. each block indicates which electron sublevel is in the process of being filled.
Comments are closed.