Difference Between Reducing And Nonreducing Sugar Definition

Difference Between Reducing And Non Reducing Sugars Learn The Key Reducing sugars contain free aldehyde or ketone groups, allowing them to undergo chemical reactions, while non reducing sugars lack these groups and remain chemically inert. Reducing sugar is a type of sugar that consists of a free aldehyde group or a free ketone group, allowing the molecule to act as a reducing agent. non reducing sugar is a type of sugar that doesn't have a free aldehyde or ketone group, as a result of which the sugar cannot act as a reducing agent.

Reducing And Non Reducing Sugar 2 Pdf Carbohydrates Chemistry The main difference between reducing and nonreducing sugar is that reducing sugars have free aldehyde or ketone groups whereas nonreducing sugars do not have free aldehyde or ketone groups. In conclusion, reducing sugars and non reducing sugars have distinct attributes that differentiate them from each other. while reducing sugars contain a free aldehyde or ketone group and exhibit reducing properties, non reducing sugars lack this functional group and do not have the same reactivity. Find a clear example of a reducing and non reducing sugar, like glucose and sucrose. learn the key differences, how to identify them, and why their properties are crucial in food and biology. What are non reducing and reducing sugars? reducing sugars contain free aldehyde or ketone groups and are capable of reducing other compounds. on the other hand, non reducing sugars do not.

Difference Between Reducing And Non Reducing Sugar Sinaumedia Find a clear example of a reducing and non reducing sugar, like glucose and sucrose. learn the key differences, how to identify them, and why their properties are crucial in food and biology. What are non reducing and reducing sugars? reducing sugars contain free aldehyde or ketone groups and are capable of reducing other compounds. on the other hand, non reducing sugars do not. Sugars can be classified as reducing or non reducing; this classification is dependent on their ability to donate electrons reducing sugars can donate electrons (the carbonyl group becomes oxidised), the sugars become the reducing agent. Reducing sugars have free carbonyl groups (either aldehyde or ketone) that can be oxidized. nonreducing sugars, however, have their carbonyl groups involved in glycosidic bonds, which prevents them from participating in oxidation reactions. Reducing sugars are carbohydrates that have free aldehyde or ketone groups, which allow them to act as reducing agents. nonreducing sugars, on the other hand, lack these free functional groups and, thus, cannot reduce other compounds. In this protocol, we are performing tests to distinguish between reducing and non reducing sugars and learn about the properties of reducing and non reducing sugars.
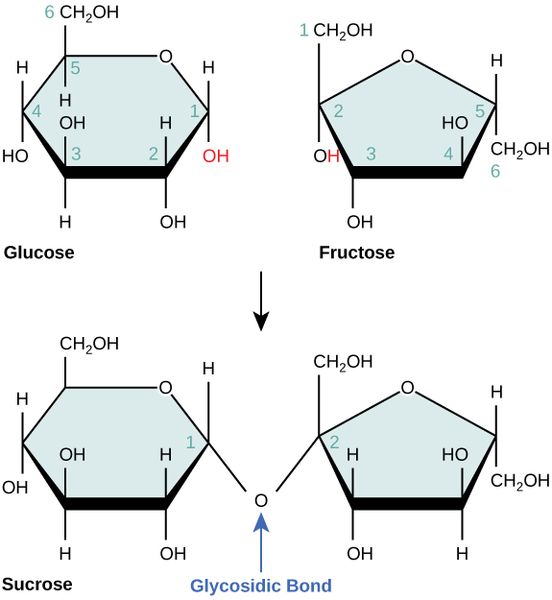
Difference Between Reducing And Nonreducing Sugar Definition Sugars can be classified as reducing or non reducing; this classification is dependent on their ability to donate electrons reducing sugars can donate electrons (the carbonyl group becomes oxidised), the sugars become the reducing agent. Reducing sugars have free carbonyl groups (either aldehyde or ketone) that can be oxidized. nonreducing sugars, however, have their carbonyl groups involved in glycosidic bonds, which prevents them from participating in oxidation reactions. Reducing sugars are carbohydrates that have free aldehyde or ketone groups, which allow them to act as reducing agents. nonreducing sugars, on the other hand, lack these free functional groups and, thus, cannot reduce other compounds. In this protocol, we are performing tests to distinguish between reducing and non reducing sugars and learn about the properties of reducing and non reducing sugars.
Comments are closed.