Diatomic Molecules
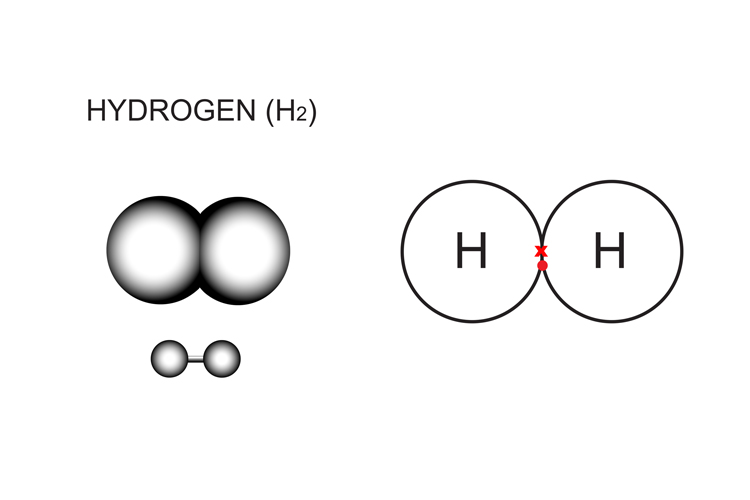
Diatomic Molecules Have Two Of The Same Element Learn about diatomic molecules, which are composed of two atoms of the same or different elements. find out their properties, occurrence, molecular geometry, and historical significance. Diatomic molecules consist of two atoms bonded together. in contrast, monatomic elements consist of single atoms (e.g., ar, he). many compounds are diatomic, such as hcl, nacl, and kbr. diatomic compounds consist of two different elements. seven pure elements form diatomic molecules.

Diatomic Molecules Over 242 Royalty Free Licensable Stock Vectors Learn what diatomic molecules are, how they are formed by chemical bonds, and which elements form them. explore the examples of homonuclear and heteronuclear diatomic molecules, such as h2, n2, o2, hcl and more. Diatomic molecules with two different atoms are called heteronuclear diatomic molecules. when two nonidentical atoms interact to form a chemical bond, the interacting atomic orbitals do not have the same energy. Diatomic molecule, any chemical compound that is made up of only two atoms. the two atoms can be the same type of atom, such as oxygen (o 2), where both atoms in the molecule are oxygen atoms; such molecules are known as homonuclear diatomic molecules. A diatomic molecule is a chemical species made up of exactly two atoms bonded together. this structural grouping classifies molecules based purely on their atomic count.

Diatomic Molecules Over 242 Royalty Free Licensable Stock Vectors Diatomic molecule, any chemical compound that is made up of only two atoms. the two atoms can be the same type of atom, such as oxygen (o 2), where both atoms in the molecule are oxygen atoms; such molecules are known as homonuclear diatomic molecules. A diatomic molecule is a chemical species made up of exactly two atoms bonded together. this structural grouping classifies molecules based purely on their atomic count. A diatomic molecule is a molecule that consists of two atoms, which can be either of the same element or different elements. these molecules are significant in chemistry because they can form covalent bonds and are fundamental in understanding molecular structures and interactions. Learn what diatomic molecules are and how they are classified into homonuclear and heteronuclear based on the identity of the atoms. find out the seven diatomic elements and their properties, and how to remember them with a mnemonic. A diatomic molecule is a molecule that consists of exactly two atoms bonded together. these atoms can either be of the same element, known as a homonuclear diatomic molecule (e.g., o₂, n₂), or of different elements, known as a heteronuclear diatomic molecule (e.g., co, hcl). This page explains the formation and bonding characteristics of heteronuclear diatomic molecules through molecular orbital theory, emphasizing concepts like polar covalent bonds, resonance, and electron configurations.
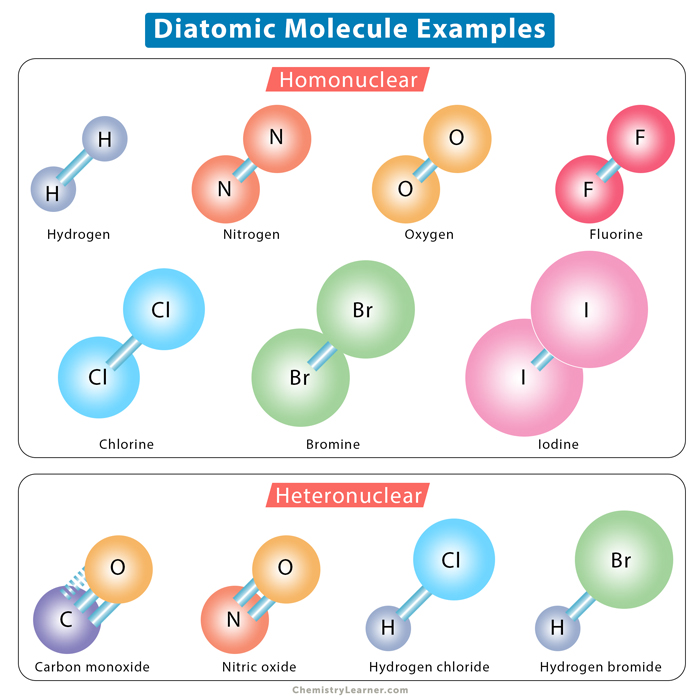
Diatomic Molecules Definition And List A diatomic molecule is a molecule that consists of two atoms, which can be either of the same element or different elements. these molecules are significant in chemistry because they can form covalent bonds and are fundamental in understanding molecular structures and interactions. Learn what diatomic molecules are and how they are classified into homonuclear and heteronuclear based on the identity of the atoms. find out the seven diatomic elements and their properties, and how to remember them with a mnemonic. A diatomic molecule is a molecule that consists of exactly two atoms bonded together. these atoms can either be of the same element, known as a homonuclear diatomic molecule (e.g., o₂, n₂), or of different elements, known as a heteronuclear diatomic molecule (e.g., co, hcl). This page explains the formation and bonding characteristics of heteronuclear diatomic molecules through molecular orbital theory, emphasizing concepts like polar covalent bonds, resonance, and electron configurations.

Diatomic Molecules Definition And List A diatomic molecule is a molecule that consists of exactly two atoms bonded together. these atoms can either be of the same element, known as a homonuclear diatomic molecule (e.g., o₂, n₂), or of different elements, known as a heteronuclear diatomic molecule (e.g., co, hcl). This page explains the formation and bonding characteristics of heteronuclear diatomic molecules through molecular orbital theory, emphasizing concepts like polar covalent bonds, resonance, and electron configurations.
Comments are closed.