Diatomic Molecule Geometry

Diatomic Molecule Alchetron The Free Social Encyclopedia For almost every covalent molecule that exists, we can now draw the lewis structure, predict the electron pair geometry, predict the molecular geometry, and come close to predicting bond angles. To do this it is necessary to classify the states of the molecule using the irreducible representations from the character table of the symmetry group of the molecule. among all the molecular symmetries, diatomic molecules show some distinct features and are relatively easier to analyze.
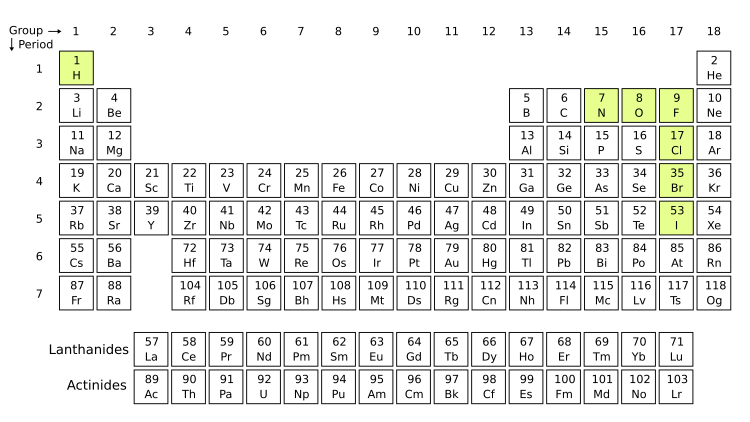
Diatomic Molecule Facts For Kids When a molecule has just one bond, it’s simple to visualize its dipole moment. once we move beyond diatomic molecules, we need to start considering how the sum of the individual dipoles (which are vectors, by the way) is affected by their arrangement in the molecule, i.e. their geometry. For the charge and multiplicity, use the data in the table below. you should run a total of six calculations since you will need to run the singlet and triplet state for each molecule. Different types of motion in a molecule happen on very different time scales. just as in clas sical mechanics this property leads to an approximate separability of the motions. conse the orientation of the molecule in space, and describes the spin state of the nuclei. (see table 3.1). In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. however, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule.

What Is A Diatomic Molecule Definition Infoupdate Org Different types of motion in a molecule happen on very different time scales. just as in clas sical mechanics this property leads to an approximate separability of the motions. conse the orientation of the molecule in space, and describes the spin state of the nuclei. (see table 3.1). In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. however, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the vsepr theory. a table of geometries using the vsepr theory can facilitate drawing and understanding molecules. Consider the schrödinger equation for a system of two identical nuclei and two electrons, in the centre of mass system of the nuclei, neglecting spin: is the separation of the nuclei, and and are the positions of the two electrons. we will solve this system by a separation and some approximations. A simple diatomic molecule consisting of two atoms of the same element will always have a linear geometry. the diatomic oxygen (o 2 ) molecule is non polar because the two oxygen atoms have the same electronegativity, and there are no partial charges on either atom. Once the molecular orbitals are calculated, the electronic configurations for the ground state of a diatomic molecule can be "filled" up from the lowest orbital and up.
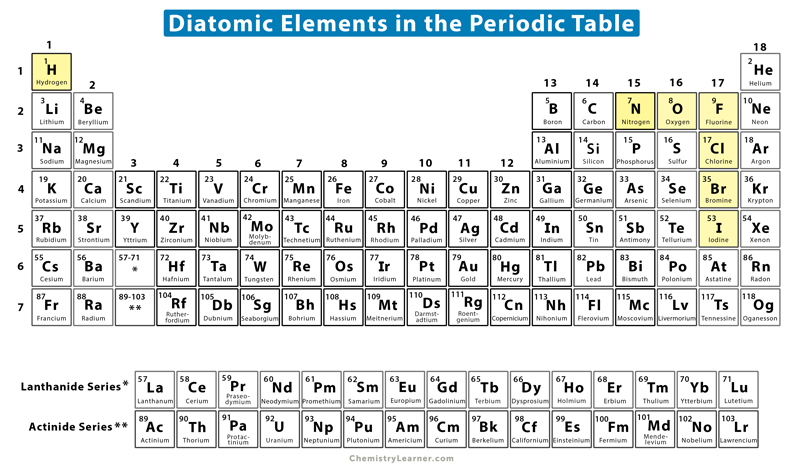
What Is A Diatomic Molecule Definition Infoupdate Org Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the vsepr theory. a table of geometries using the vsepr theory can facilitate drawing and understanding molecules. Consider the schrödinger equation for a system of two identical nuclei and two electrons, in the centre of mass system of the nuclei, neglecting spin: is the separation of the nuclei, and and are the positions of the two electrons. we will solve this system by a separation and some approximations. A simple diatomic molecule consisting of two atoms of the same element will always have a linear geometry. the diatomic oxygen (o 2 ) molecule is non polar because the two oxygen atoms have the same electronegativity, and there are no partial charges on either atom. Once the molecular orbitals are calculated, the electronic configurations for the ground state of a diatomic molecule can be "filled" up from the lowest orbital and up.

Linear Molecular Geometry Diatomic Molecule Vsepr Theory Png Clipart A simple diatomic molecule consisting of two atoms of the same element will always have a linear geometry. the diatomic oxygen (o 2 ) molecule is non polar because the two oxygen atoms have the same electronegativity, and there are no partial charges on either atom. Once the molecular orbitals are calculated, the electronic configurations for the ground state of a diatomic molecule can be "filled" up from the lowest orbital and up.

Electron Geometry Chart Diatomic Hopefity
Comments are closed.