Diatomic Elements Pdf

Metals Diatomic Elements Hydrogen H Pdf Diatomic elements free download as word doc (.doc .docx), pdf file (.pdf), text file (.txt) or read online for free. this document lists diatomic elements and metals that form diatomic molecules or ions. While some elements exist as molecules, most elements exist with individual atoms as their basic unit. if an element is not one of those listed above as a molecular element, assume it is an atomic element.

Atomicity Monoatomic Diatomic Triatomic Polyatomic Ions And Diatomic elements only exist in nature as a pair of atoms of the same element that are bonded together. the seven diatomic elements are h2, n2, o2, f2, cl2, br2, and i2. All atoms of a given element are identical, having the same size, mass and chemical properties. the atoms of one element are different from the atoms of all other elements. Diatomic and polyatomic elements to memorize. Chapter 6 – chemical bonding diatomic molecules & lewis structures diatomic molecules include: h2, n2, o2, f2, cl2, br2, or i2.
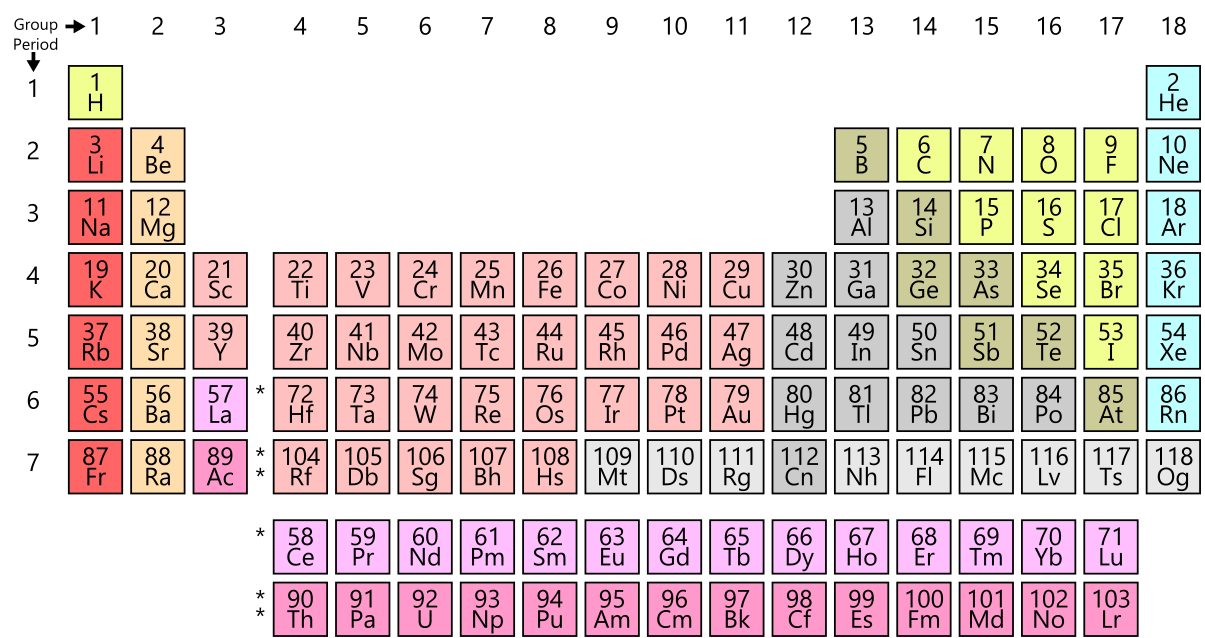
Diatomic Elements Science Trends Diatomic and polyatomic elements to memorize. Chapter 6 – chemical bonding diatomic molecules & lewis structures diatomic molecules include: h2, n2, o2, f2, cl2, br2, or i2. If the material is a pure substance, further classify it as either an element or compound in the right column. similarly, if the material is a mixture, further classify it as homogeneous or heterogeneous in the right column. Chemistry is my jam use the periodic table below to indicate the elements that are diatomic. true or false: carbon is more stable bonded to another atom of carbon then it is alone. a diatomic element. it will always have a “2” for a subscript in. Diatomic molecules molecules that have two atoms are called diatomic. hydrogen (h2), nitrogen (n2), oxygen (o2), fluorine (f2), chlorine (cl2), bromine (br2), and iodine (i2) are diatomic. All atoms of a given element are identical, having the same size, mass and chemical properties. the atoms of one element are different from the atoms of all other elements.

Diatomic Elements Pdf If the material is a pure substance, further classify it as either an element or compound in the right column. similarly, if the material is a mixture, further classify it as homogeneous or heterogeneous in the right column. Chemistry is my jam use the periodic table below to indicate the elements that are diatomic. true or false: carbon is more stable bonded to another atom of carbon then it is alone. a diatomic element. it will always have a “2” for a subscript in. Diatomic molecules molecules that have two atoms are called diatomic. hydrogen (h2), nitrogen (n2), oxygen (o2), fluorine (f2), chlorine (cl2), bromine (br2), and iodine (i2) are diatomic. All atoms of a given element are identical, having the same size, mass and chemical properties. the atoms of one element are different from the atoms of all other elements.

What Are The 7 Diatomic Elements Definition And List Diatomic molecules molecules that have two atoms are called diatomic. hydrogen (h2), nitrogen (n2), oxygen (o2), fluorine (f2), chlorine (cl2), bromine (br2), and iodine (i2) are diatomic. All atoms of a given element are identical, having the same size, mass and chemical properties. the atoms of one element are different from the atoms of all other elements.
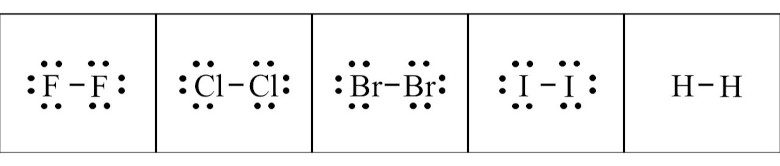
Diatomic Elements Easy Hard Science
Comments are closed.