Diatomic Elements Molecules
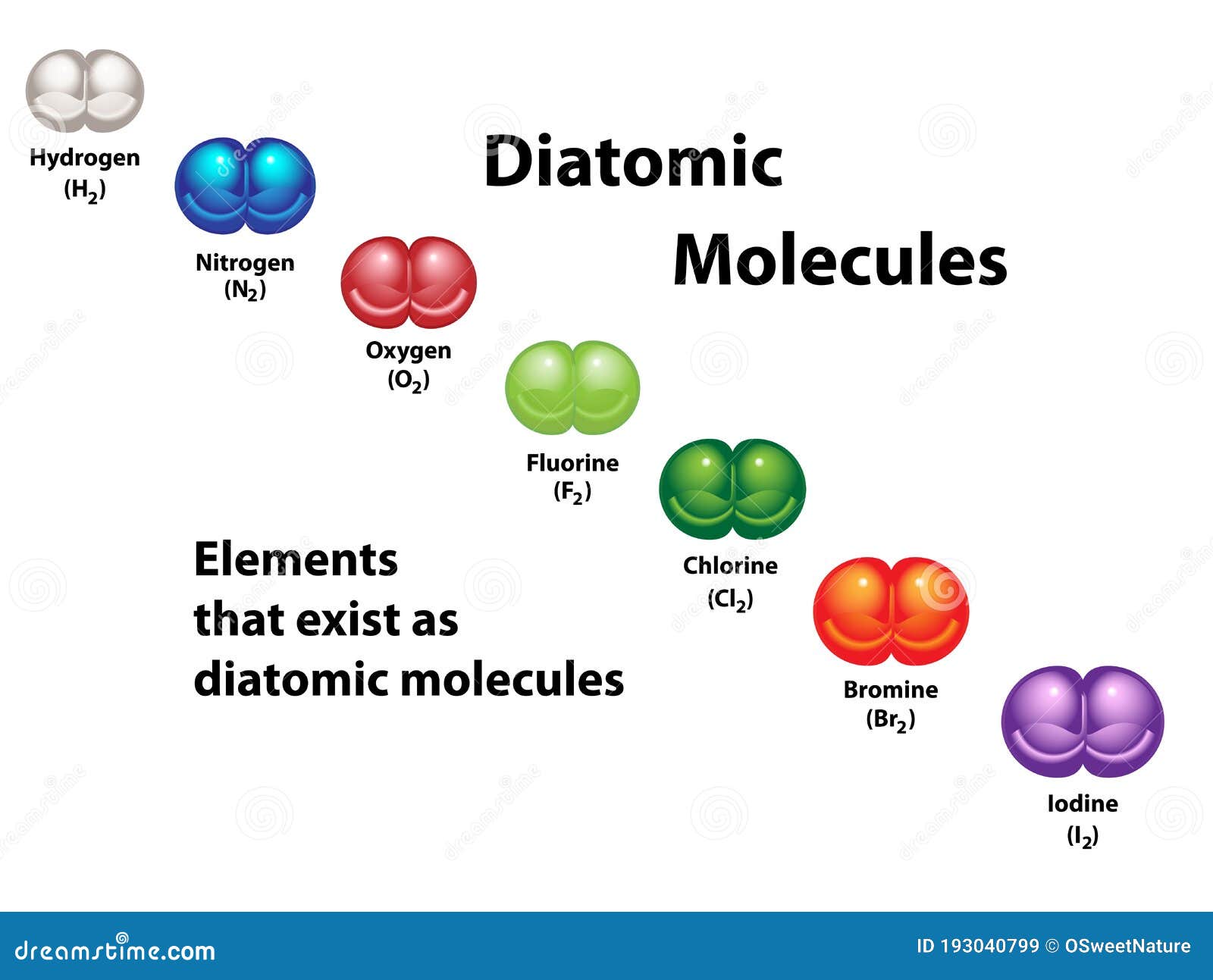
Diatomic Molecules Elements Diagram Colors Stock Vector Illustration Diatomic elements are pure elements that form molecules consisting of two atoms bonded together. there are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, and bromine. There are seven diatomic elements, aka molecular elements, all listed here. learn about what a diatomic element is and how it's different from a diatomic molecule.
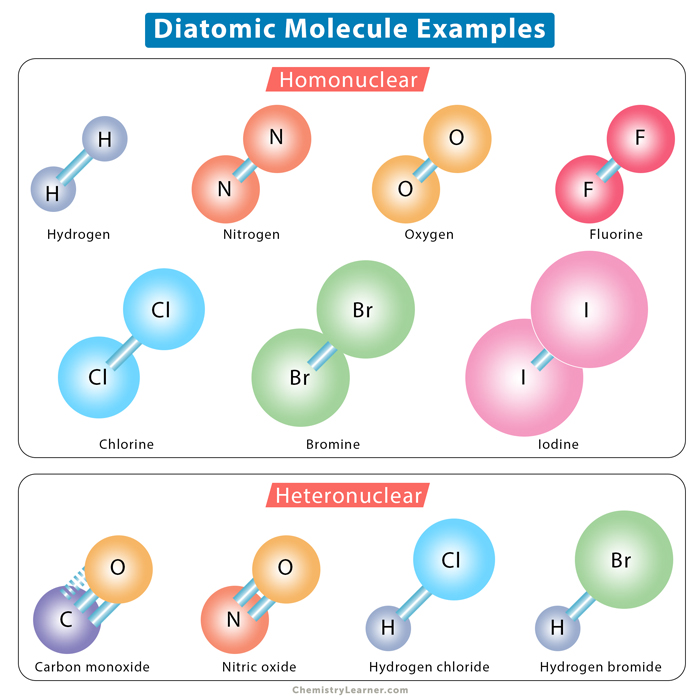
Diatomic Molecules Definition And List While some elements exist as molecules, most elements exist with individual atoms as their basic unit. if an element is not one of those listed above as a molecular element, assume it is an atomic element. Diatomic molecules (from greek di 'two') are molecules composed of only two atoms, of the same or different chemical elements. if a diatomic molecule consists of two atoms of the same element, such as hydrogen (h2) or oxygen (o2), then it is said to be homonuclear. While diatomic elements are uncommon, diatomic molecules readily occur. common examples include sodium chloride (nacl), carbon monoxide (co), and nitric oxide (no). Diatomic molecule, any chemical compound that is made up of only two atoms. the two atoms can be the same type of atom, such as oxygen (o2), where both atoms in the molecule are oxygen atoms; such molecules are known as homonuclear diatomic molecules.

Diatomic Molecules Examples While diatomic elements are uncommon, diatomic molecules readily occur. common examples include sodium chloride (nacl), carbon monoxide (co), and nitric oxide (no). Diatomic molecule, any chemical compound that is made up of only two atoms. the two atoms can be the same type of atom, such as oxygen (o2), where both atoms in the molecule are oxygen atoms; such molecules are known as homonuclear diatomic molecules. Because the number of atoms in a molecule defines the prefix, a molecule containing two atoms is called diatomic. the prefix di , which means “two,” is greek in origin. a homonuclear diatomic molecule is generated by the chemical reaction of two atoms of equivalent type. Learn what diatomic molecules are, which elements form them, and the chemical stability that drives two atoms to bond. What are diatomic molecules and diatomic elements. learn homonuclear and heteronuclear diatomic molecules with examples and diagrams. how to remember them. Additionally, some elements pair up as diatomic molecules, such as o₂ and h₂, rather than existing as single atoms. understanding whether a substance is molecular or crystalline and how diatomic elements differ from compounds is key to classifying matter.

Diatomic Molecules Examples Because the number of atoms in a molecule defines the prefix, a molecule containing two atoms is called diatomic. the prefix di , which means “two,” is greek in origin. a homonuclear diatomic molecule is generated by the chemical reaction of two atoms of equivalent type. Learn what diatomic molecules are, which elements form them, and the chemical stability that drives two atoms to bond. What are diatomic molecules and diatomic elements. learn homonuclear and heteronuclear diatomic molecules with examples and diagrams. how to remember them. Additionally, some elements pair up as diatomic molecules, such as o₂ and h₂, rather than existing as single atoms. understanding whether a substance is molecular or crystalline and how diatomic elements differ from compounds is key to classifying matter.
Comments are closed.