Diatomic Elements Easy Hard Science
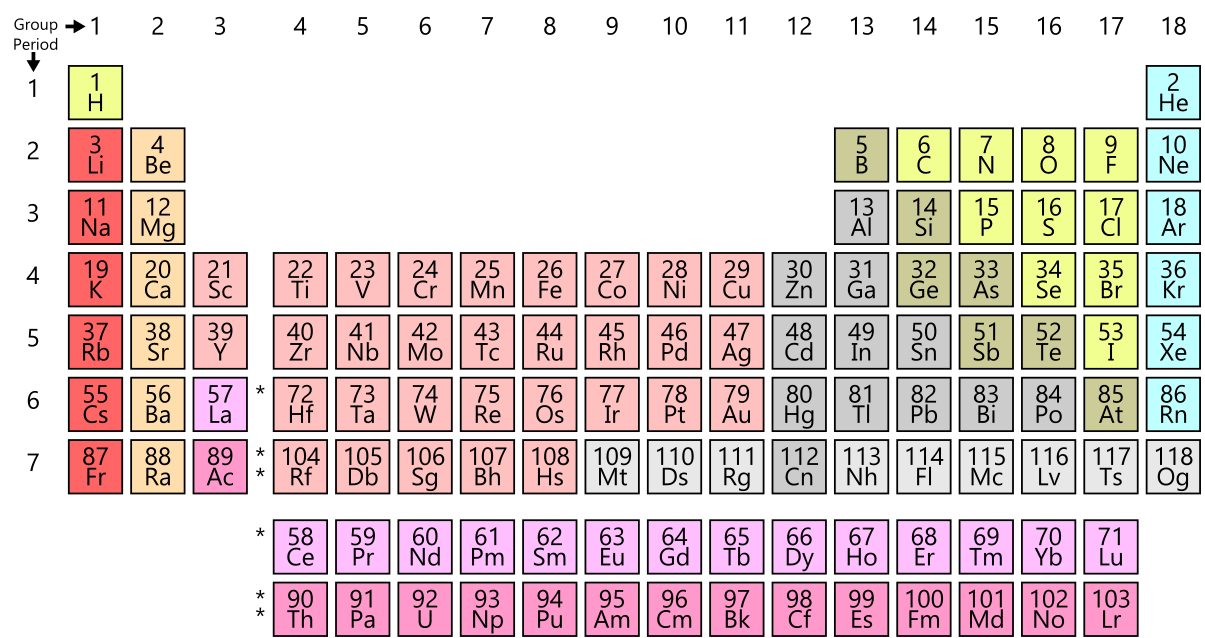
Diatomic Elements Science Trends The 7 diatomic elements are hydrogen (h), nitrogen (n), oxygen (o), fluorine (f), chlorine (cl), bromine (br), and iodine (i). we call them diatomic elements because the atoms appear in pairs. Diatomic elements are pure elements that form molecules consisting of two atoms bonded together. there are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, and bromine.

What Are The 7 Diatomic Elements Definition And List Learn about diatomic molecules with simple definitions, facts, examples, and interactive quizzes. perfect for middle school science students. There are seven diatomic elements, aka molecular elements, all listed here. learn about what a diatomic element is and how it's different from a diatomic molecule. Being able to remember and identify diatomic elements is essential to success in chemistry! in this video we explain why the diatomic elements exist and give you an easy trick to. Diatomic molecules are tiny particles (called molecules) made of only two atoms. these two atoms can be the same kind of chemical element or different kinds. if a diatomic molecule has two atoms of the same element, like hydrogen (h 2) or oxygen (o 2), it's called a homonuclear molecule.
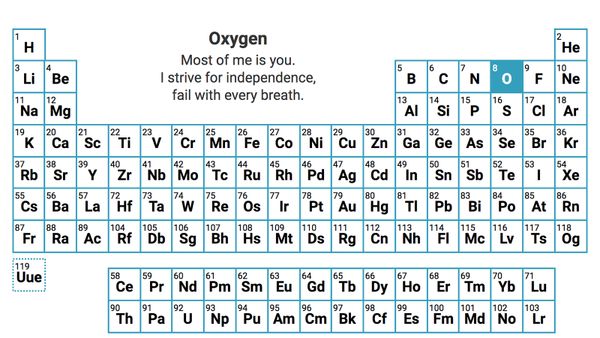
The 7 Diatomic Elements That Can T Stand To Be Alone Howstuffworks Being able to remember and identify diatomic elements is essential to success in chemistry! in this video we explain why the diatomic elements exist and give you an easy trick to. Diatomic molecules are tiny particles (called molecules) made of only two atoms. these two atoms can be the same kind of chemical element or different kinds. if a diatomic molecule has two atoms of the same element, like hydrogen (h 2) or oxygen (o 2), it's called a homonuclear molecule. The formulae for most elements is just their symbol. the exceptions are some non metal elements known as diatomic elements . they contain two atoms of the same element covalently bonded. Diatomic elements are molecules composed of only two atoms, every time, always. there are only seven of them on the entire periodic table. Understanding whether a substance is molecular or crystalline and how diatomic elements differ from compounds is key to classifying matter. created by mahesh shenoy. This is a list of all of the diatomic elements and their common properties. simple mnemonics for remembering them are included.
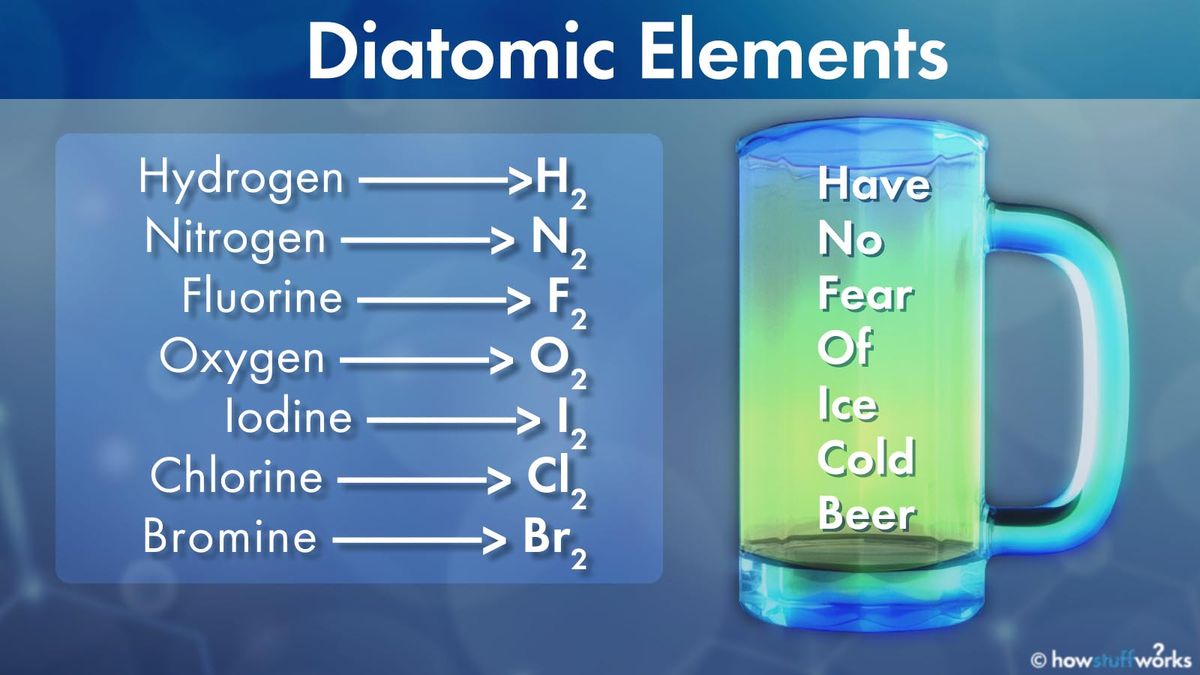
The 7 Diatomic Elements That Can T Stand To Be Alone Howstuffworks The formulae for most elements is just their symbol. the exceptions are some non metal elements known as diatomic elements . they contain two atoms of the same element covalently bonded. Diatomic elements are molecules composed of only two atoms, every time, always. there are only seven of them on the entire periodic table. Understanding whether a substance is molecular or crystalline and how diatomic elements differ from compounds is key to classifying matter. created by mahesh shenoy. This is a list of all of the diatomic elements and their common properties. simple mnemonics for remembering them are included.
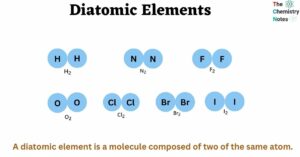
Diatomic Elements Important 7 Elements Properties Formation Understanding whether a substance is molecular or crystalline and how diatomic elements differ from compounds is key to classifying matter. created by mahesh shenoy. This is a list of all of the diatomic elements and their common properties. simple mnemonics for remembering them are included.
Comments are closed.