Diatomic Elements

The Diatomic Elements Chemtalk Diatomic elements are pure elements that form molecules consisting of two atoms bonded together. there are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, and bromine. Learn what diatomic elements are and which ones exist at ordinary conditions. find out how to remember the seven diatomic elements (h, n, o, f, cl, br, i) with easy mnemonics.

The Diatomic Elements Chemtalk Learn what diatomic elements are, how they differ from diatomic molecules, and how to remember the seven diatomic elements: hydrogen, nitrogen, fluorine, oxygen, iodine, bromine, and chlorine. find out their properties, uses, and examples of diatomic molecules. A diatomic molecule is composed of two atoms, of the same or different elements. learn about the types, properties, occurrence, and history of diatomic molecules, such as hydrogen, oxygen, nitrogen, and carbon monoxide. Learn what diatomic elements are, how to write their formulas, and which elements are diatomic. also, find out which diatomic elements are gases, liquids, or solids at standard conditions. Learn what diatomic elements are, how they form, and which seven elements are diatomic in nature. find out the physical and chemical properties of hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine.

What Are The 7 Diatomic Elements Definition And List Learn what diatomic elements are, how to write their formulas, and which elements are diatomic. also, find out which diatomic elements are gases, liquids, or solids at standard conditions. Learn what diatomic elements are, how they form, and which seven elements are diatomic in nature. find out the physical and chemical properties of hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine. Learn what diatomic molecules are and how to distinguish between homonuclear and heteronuclear types. find out the seven diatomic elements and their properties, and how to remember them with a mnemonic. Learn what diatomic elements are, how they are bonded, and where they are located on the periodic table. find out the seven diatomic elements and their acronym, and watch a video lesson on the topic. Learn about the 7 diatomic elements (h, n, o, f, cl, br, i) and their structures, nomenclature, and properties. find out why they are diatomic and how to name them correctly. Learn what diatomic elements are, how to remember them and why they are so common in the atmosphere and chemistry. find out the names, symbols and properties of the seven diatomic elements: hydrogen, nitrogen, fluorine, oxygen, iodine, chlorine and bromine.
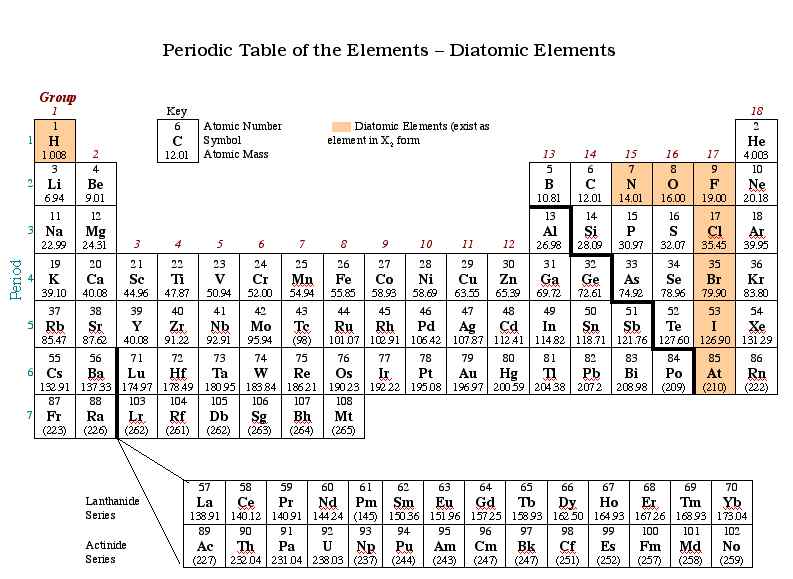
1b6 Diatomic Elements Learn what diatomic molecules are and how to distinguish between homonuclear and heteronuclear types. find out the seven diatomic elements and their properties, and how to remember them with a mnemonic. Learn what diatomic elements are, how they are bonded, and where they are located on the periodic table. find out the seven diatomic elements and their acronym, and watch a video lesson on the topic. Learn about the 7 diatomic elements (h, n, o, f, cl, br, i) and their structures, nomenclature, and properties. find out why they are diatomic and how to name them correctly. Learn what diatomic elements are, how to remember them and why they are so common in the atmosphere and chemistry. find out the names, symbols and properties of the seven diatomic elements: hydrogen, nitrogen, fluorine, oxygen, iodine, chlorine and bromine.
Comments are closed.