Device Classification In India Infographic
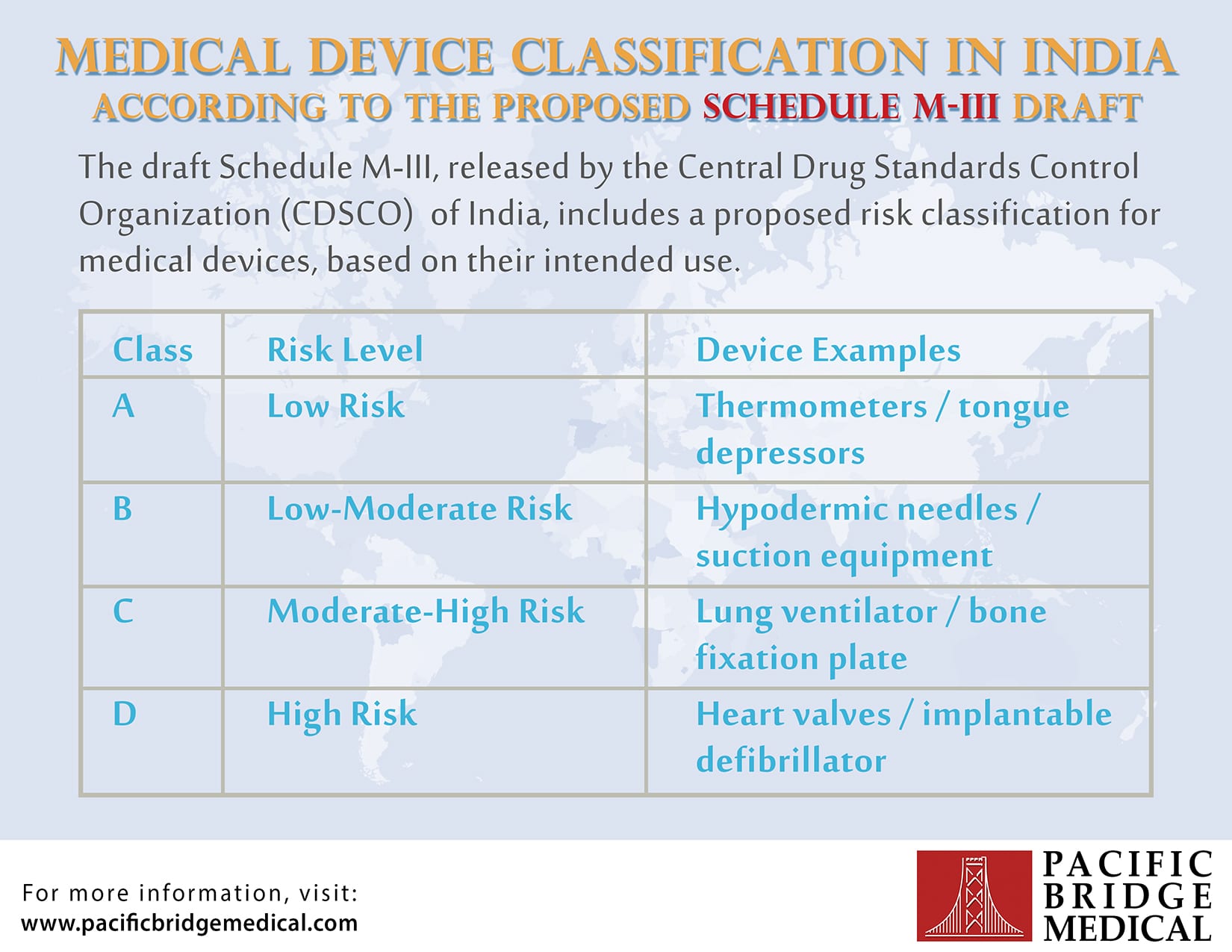
Device Classification In India Infographic Diagnostic x ray beam limiting device is a device such as a collimator, a cone, or an aperture intended to restrict the dimensions of a diagnostic x ray field by limiting the size of the primary x ray beam. The central drug standards control organization (cdsco) released a draft document, schedule m iii, which contains proposed official definitions for various types of medical devices in india.

Medical Device Classification India At Stephanie Post Blog Learn about cdsco classification for medical devices in india, including device categories, regulatory pathways, and compliance for market approval. This review presents a detailed comparative analysis of classification systems across the united states (fda), european union (eu mdr), japan (pmda), canada (health canada), australia (tga), china (nmpa), and india (cdsco). Increased health awareness, a growing middle class, and government health efforts are projected to propel india's medical equipment market forward in the next years. with the publication of the. Explore india's risk based classification for medical devices and ivds. learn about regulatory categories, compliance requirements, and risk levels from class a to class d.

Medical Device Classification India At Stephanie Post Blog Increased health awareness, a growing middle class, and government health efforts are projected to propel india's medical equipment market forward in the next years. with the publication of the. Explore india's risk based classification for medical devices and ivds. learn about regulatory categories, compliance requirements, and risk levels from class a to class d. Learn about the classification and types of medical devices as defined by cdsco. understand the differences between notified and non notified devices. This tutorial provides a comprehensive guide on the classification of medical devices into four classes: a, b, c, and d, along with associated regulatory requirements and compliance actions. These rules cover various aspects of device related regulations, including classification, registration, manufacturing and import, labeling, sales, and postmarket requirements, etc. Class b system is a closed cycle cryosurgical device intended to ablate the endometrial lining of the uterus in premenopausal women with menorrhagia (excessive bleeding) due to benign causes for whom childbearing is complete. it is intended for intracardiac ablation.

Medical Device Classification India At Stephanie Post Blog Learn about the classification and types of medical devices as defined by cdsco. understand the differences between notified and non notified devices. This tutorial provides a comprehensive guide on the classification of medical devices into four classes: a, b, c, and d, along with associated regulatory requirements and compliance actions. These rules cover various aspects of device related regulations, including classification, registration, manufacturing and import, labeling, sales, and postmarket requirements, etc. Class b system is a closed cycle cryosurgical device intended to ablate the endometrial lining of the uterus in premenopausal women with menorrhagia (excessive bleeding) due to benign causes for whom childbearing is complete. it is intended for intracardiac ablation.

Medical Device Classification India At Stephanie Post Blog These rules cover various aspects of device related regulations, including classification, registration, manufacturing and import, labeling, sales, and postmarket requirements, etc. Class b system is a closed cycle cryosurgical device intended to ablate the endometrial lining of the uterus in premenopausal women with menorrhagia (excessive bleeding) due to benign causes for whom childbearing is complete. it is intended for intracardiac ablation.
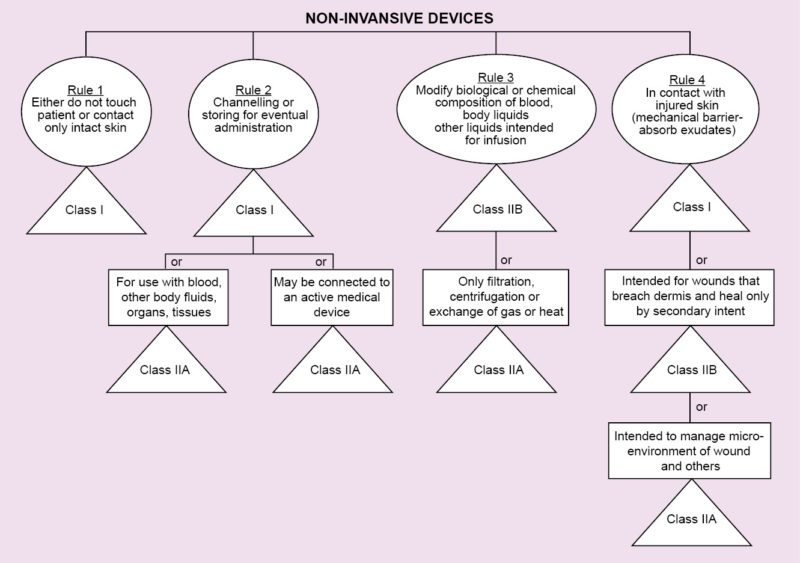
Medical Device Classification India At Stephanie Post Blog
Comments are closed.