Design Controls Process Design Talk

Design Controls Process Flow Pdf Design control refers to the set of technical requirements and procedures followed during the design and development process of a device to ensure it meets customer, technical, and regulatory expectations, while also managing project costs by minimizing unnecessary design changes. Design reviews take place at the various stages of the design control procedures. during the design review meetings, the progress made on each of the design stages is reviewed in detail to make the device is safe and design control parameters are being implemented consistently and properly.
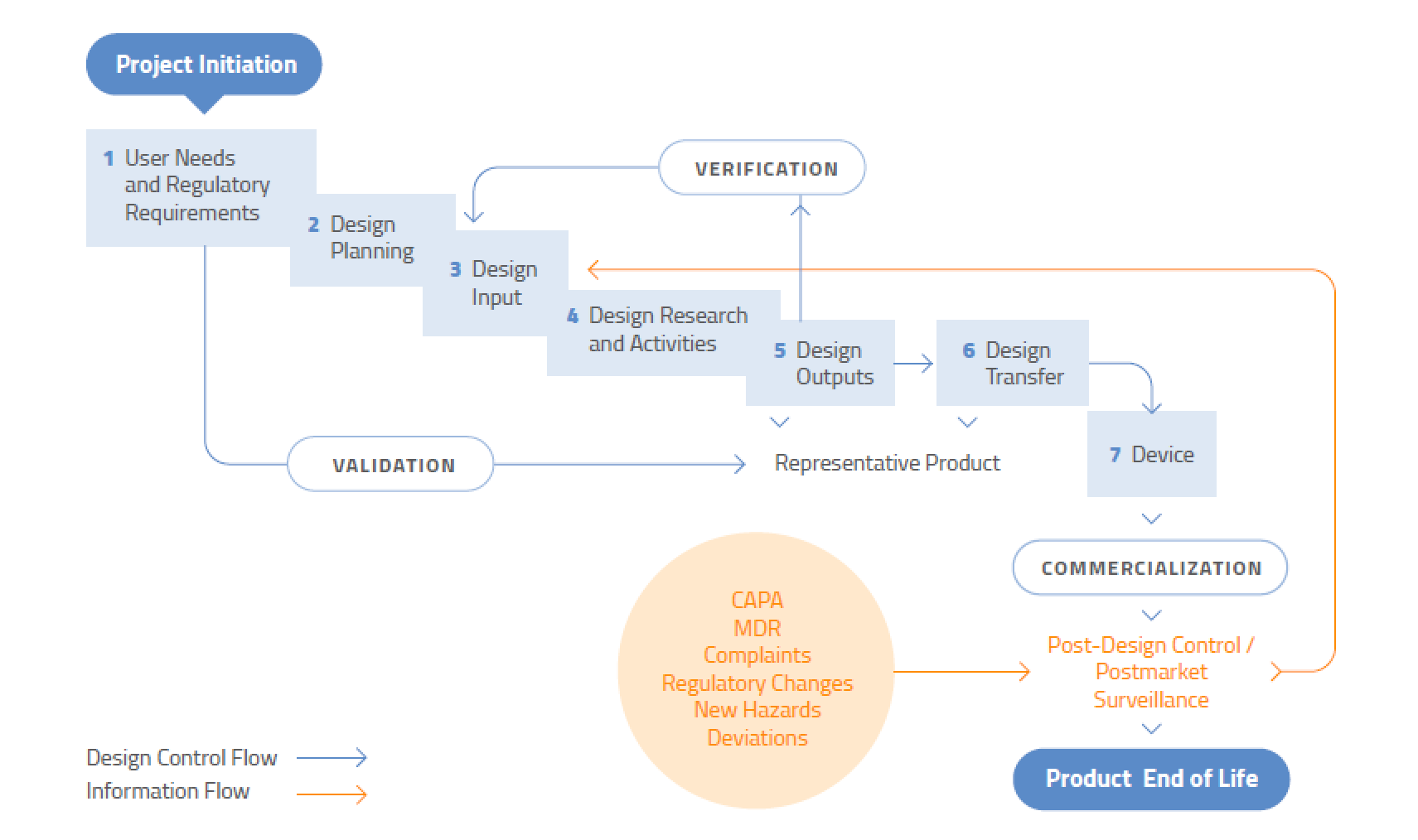
Design Controls Process Design Talk In 60 minutes, towler explains the basics of process control and discusses how and when to apply them in your process design work. you’ll learn how to read and understand a p&id diagram and put together a basic process control scheme. What? design controls are an interrelated set of quality practices and procedures that are incorporated into the design and development process of a medical device. Aimed at chemical engineering students in process control courses, as well as practicing process and control engineers, this textbook offers an alternative to traditional texts and offers a practical, hands on approach to design of process controls. At its core, process control design involves the careful selection and arrangement of control elements, including sensors, controllers, actuators, and feedback mechanisms, to create a cohesive system that can respond to variations and disturbances while maintaining optimal performance.
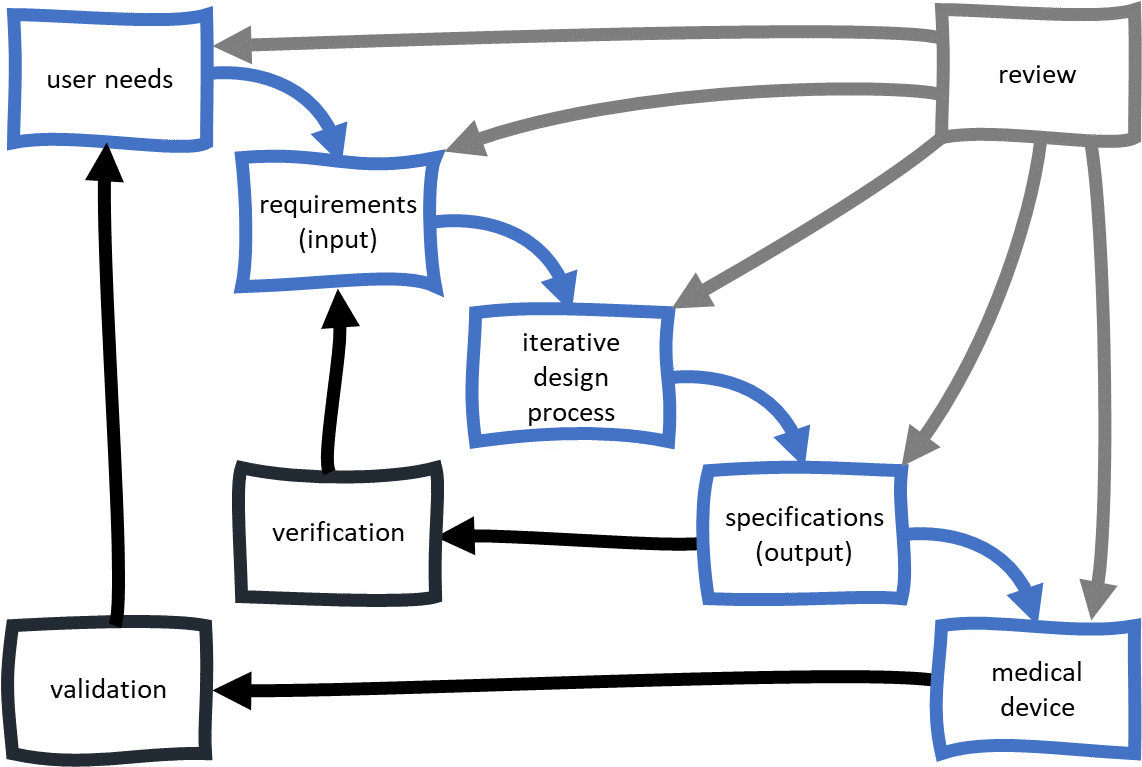
Design Controls Process Design Talk Aimed at chemical engineering students in process control courses, as well as practicing process and control engineers, this textbook offers an alternative to traditional texts and offers a practical, hands on approach to design of process controls. At its core, process control design involves the careful selection and arrangement of control elements, including sensors, controllers, actuators, and feedback mechanisms, to create a cohesive system that can respond to variations and disturbances while maintaining optimal performance. Aimed at chemical engineering students in process control courses, as well as practicing process and control engineers, this textbook offers an alternative to traditional texts and offers a. The first step to designing or analyzing a process control scheme is to determine the various inputs and outputs. there are three broad categories for any given system: inputs, outputs, and constants or parameters. Design controls take a risk based approach and the document rigor increases with product risk. design controls apply to all class i, class ii and class iii samd devices and all other class ii and class iii devices. product class within the fda database will help determine classification. This chapter provides a high level introduction to process control by starting in sect.1.1 with a brief explanation on why and when process control is needed, where and how process control fits in.
Comments are closed.