Ctops Studyx Usdm Digital Protocol Agentic Platform Clinical Protocol Automation

Mountain Protocol Usd Usdm Price Chart Related Dapps Projects Built on usdm aligned architecture and powered by agentic ai, ctops studyx enables clinical trial stakeholders—sponsors, cros, sites, and health authorities—to collaborate seamlessly, ensuring faster, more accurate protocol reviews. Welcome to ctops study x, an agentic usdm digital protocol review and publishing platform designed to transform how clinical protocols are authored, reviewed, validated, and.

Navigating Toward A Digital Clinical Trial Protocol Ctops studyx usdm digital protocol agentic platform is an ai powered digital solution for clinical protocol automation. designed for sponsors, cros, and site users, it. Learn how a hyper growth biotech replaced manual spreadsheets with custom veeva vault workflows to scale etmf completeness during rapid clinical trials. most life sciences companies cannot describe what data they have, where it lives, or what can be done with it. Mushroom solutions, a trailblazer in innovative healthcare and life sciences solutions, proudly announces the launch of its clinical trials operations automation solutions (ctops). The mushroom solutions team took center stage at the amg world – disruptive technology for clinical trials 2024 event in boston, ma, where we presented our revolutionary ctops – clinical.
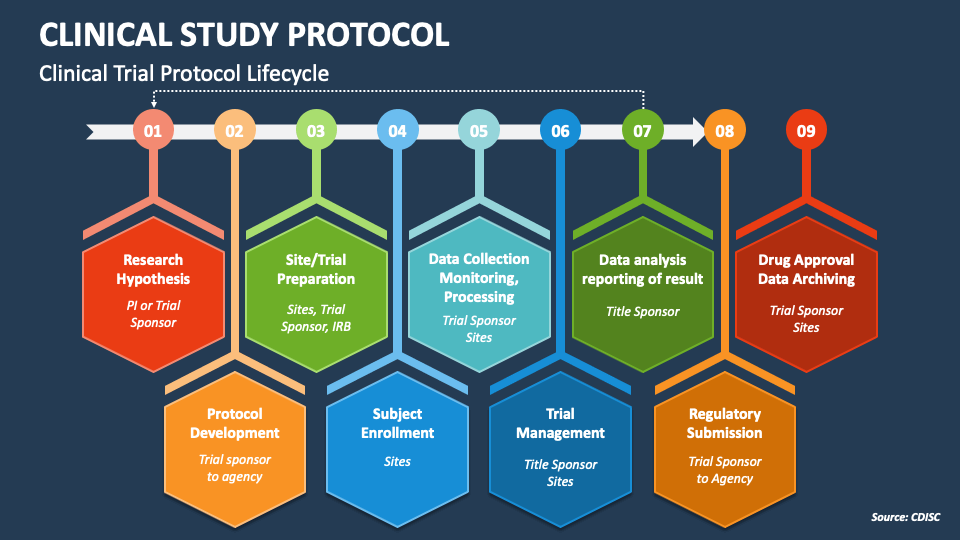
Clinical Study Protocol Powerpoint And Google Slides Template Ppt Slides Mushroom solutions, a trailblazer in innovative healthcare and life sciences solutions, proudly announces the launch of its clinical trials operations automation solutions (ctops). The mushroom solutions team took center stage at the amg world – disruptive technology for clinical trials 2024 event in boston, ma, where we presented our revolutionary ctops – clinical. Digital data flow initiative will help modernize clinical trials by enabling a digital workflow with protocol digitization. this initiative establishes a foundation for a future state of automated & dynamic readiness that can transform the drug development process. Linking the protocol to bcs to generate data contracts provides the capabilities that has been discussed in the industry for, at least, the last decade: end to end study data automation. The implementation of an electronic protocol (eprotocol) in clinical trials is set to revolutionize the entire process by enabling end to end automation, from study design to submission to regulatory authorities. The primary focus was on mapping clinical trial protocols to site feasibility management workflows using data from the shared investigator platform (sip). key deliverables included an ontology lexicon, owl files, documentation, and a proof of concept (poc) demo.
Comments are closed.