Crystalline Solids Chm361

Diagram Of Crystalline Solids Quizlet In this video, we break down the fundamental structures of crystalline solids, explore unit cells, and explain how microscopic bonding dictates macroscopic properties. This tutorial covers the fundamentals of crystalline solids, including unit cell structures, crystal arrangements of compounds like nacl and cscl, and the differences between diamond and graphite.
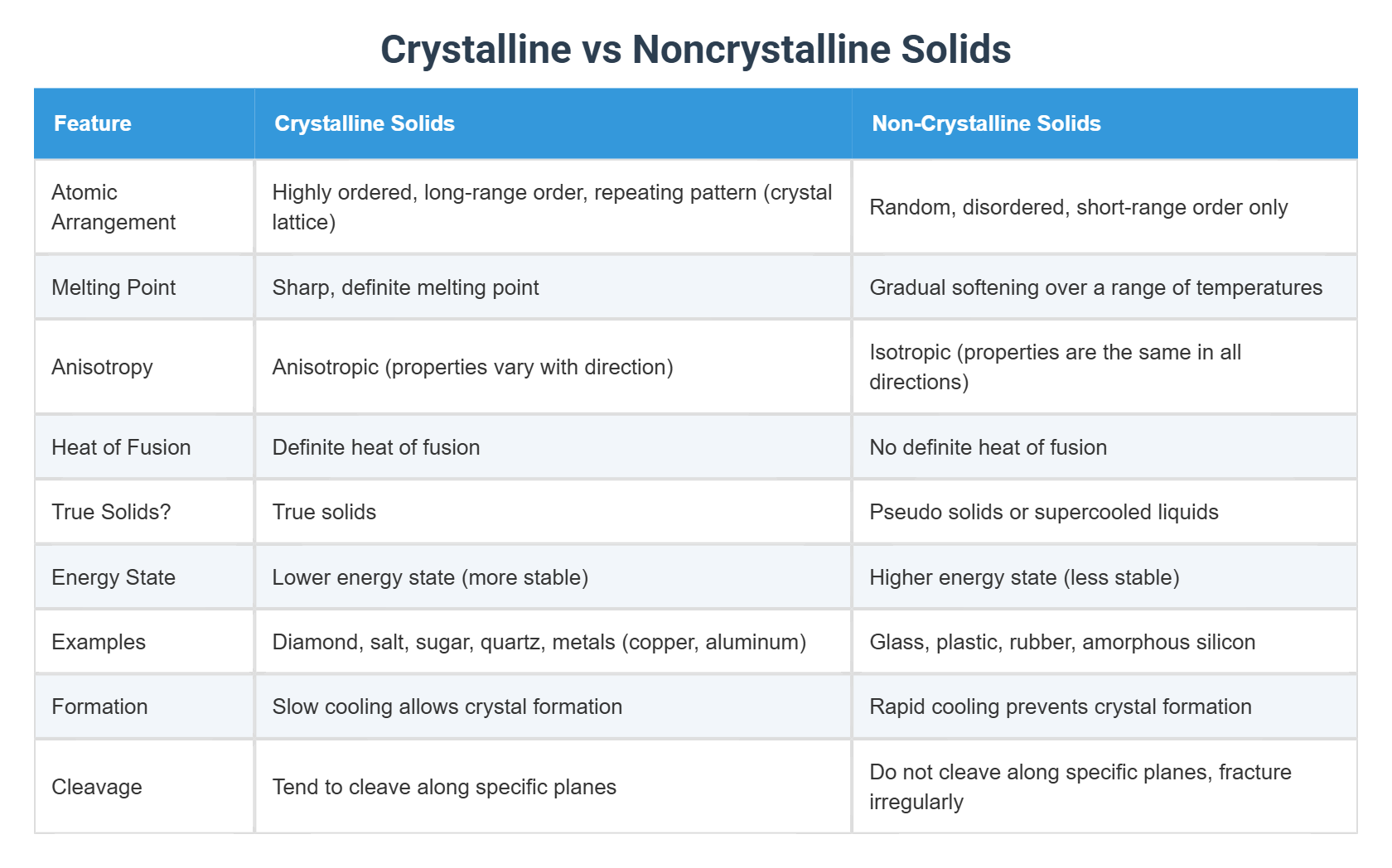
Crystalline Vs Noncrystalline Solids 2) draw the crystal structure of: a) nacl b) cscl 3) a compound formed by elements x and y crystallizes in the cubic structure where y are at the corners of the cube and x atoms are at the alternate faces. what is the formula of the compound? 4) differentiate between diamond and graphite. Chm361 tutorial 3 free download as pdf file (.pdf), text file (.txt) or read online for free. practice!. This course will provide knowledge in valence bond theory and orbital hybridization, molecular orbital theory, crystalline and solid state, transition metals, coordination compounds and metallurgy of iron. Such treatment will lead to the formation of a solid precipitate composed of insoluble calcium hydroxide. the sodium hydroxide, on the other hand, remains dissolved in the water. the solid containing calcium ions can be separated from the aqueous solution mixed with sodium ions using filtration.

Non Crystalline Solids Term This course will provide knowledge in valence bond theory and orbital hybridization, molecular orbital theory, crystalline and solid state, transition metals, coordination compounds and metallurgy of iron. Such treatment will lead to the formation of a solid precipitate composed of insoluble calcium hydroxide. the sodium hydroxide, on the other hand, remains dissolved in the water. the solid containing calcium ions can be separated from the aqueous solution mixed with sodium ions using filtration. ≥97% (hplc), crystalline solid, enos inhibitor, calbiochem® cas number: 4673 26 1; synonyms: diphenyleneiodonium chloride,dpi at sigma aldrich. Oxalic acid is suitable to be used because it is available in solid form so it is not hygroscopic. the titration was stopped once a permanent pink colour was produced. Crystalline solids, or crystals, have distinctive internal structures that in turn lead to distinctive flat surfaces, or faces. the faces intersect at angles that are characteristic of the substance. In this article, we will understand the meaning of crystalline solids, its types, properties and applications, the difference between crystalline solids and amorphous solids, and the meaning of crystal lattices.
Comments are closed.